the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Towards a more integrative approach for environmental decision-making in Brazilian transitional waters: improving biomonitoring surveys with a benthic foraminiferal biotic index
Vincent M. P. Bouchet
Silvia Helena de Mello e Sousa
Carla Bonetti
Leticia Burone
Pierre Belart
Wania Duleba
Fabio Francescangeli
Fabrizio Frontalini
Lazaro Laut
Débora S. Raposo
André R. Rodrigues
Sibelle Trevisan Disaró
Daniel Vicente Pupo
Fabrício Leandro Damasceno
Jean-Charles Pavard
Maria Virgínia Alves Martins
This study represents the first attempt to determine the indicator values of benthic foraminiferal species in Brazilian transitional waters. It also uses a regionally adapted species list to explore the potential application of Foram-AMBI, a biotic index for ecological quality. To test this, we assigned 95 living (rose-bengal-stained) benthic foraminiferal species into five ecological groups (EGs), based on the weighted-averaging (WA) optimum and tolerance to the total organic carbon (TOC) contents. Selected and published regional studies were used as the database, while independent Brazilian datasets from Sepetiba Bay and Guanabara Bay – the most polluted regions – were used to validate the ecological group assignments through Foram-AMBI. Furthermore, ecological quality status (EcoQS) criteria adapted to Brazil were developed for Foram-AMBI. The index accurately reflects the degraded environmental conditions in these two ecosystems, with moderate to poor ecological quality status in the most polluted areas. This was further confirmed by significant correlations between Foram-AMBI and TOC in both bays. This study highlights the importance of developing regional species lists and EcoQS criteria for Foram-AMBI, as the accuracy of the Brazilian list was better than that of the European list. While further research across broader pollution gradients is needed, our findings confirm the suitability and reliability of benthic foraminifera as biological indicators for assessing environmental quality in transitional waters.
- Article
(6465 KB) - Full-text XML
- BibTeX
- EndNote
Brazil hosts the second-largest mangrove area in the world, which accounts for 13.5 % of the annually sequestered carbon in the world's mangroves (Rovai et al., 2022). In addition, blue carbon storage in the woody biomass of Brazilian mangroves represents 10 % of the carbon sequestration in mangrove woody biomass globally (Rovai et al., 2022). Unfortunately, coastal areas, with their high population density and associated activities, are often subject to major human impacts (Diaz and Rosenberg, 2008). Rapid urban development and industrialisation have directly promoted the widespread contamination of Brazilian coastal areas. Accordingly, solid urban waste, industrial effluents, and micro- and nanoplastics, among other factors, have altered the ecological quality, impaired ecological functions, and led to a loss in ecosystem goods and services (e.g. Baptista Neto et al., 2006; Souza et al., 2021a). These areas, like those of southeastern Brazilian bays, have also been negatively affected by the discharges of metals, hydrocarbons, and other organic compounds, resulting in a decline in water and sediment quality (e.g. Baptista Neto et al., 2000; Ribeiro et al., 2015).
In Brazil, the National Environment Council (CONAMA), subordinated to the Ministry of the Environment and Climate Change, is responsible for defining the guidelines and standards for the environmental monitoring of ecosystems, including coastal and marine areas (Brazil, 1981). The CONAMA Resolution 357/05 (Brazil, 2005) defines the water classes, uses, and quality standards for major pollutants. It also foresees the use of “biological indicators, when appropriate, and through the use of aquatic organisms and/or communities” to evaluate the quality of aquatic environments, thus formalising the legal framework to conduct an environmental survey based on biota. Benthic macro-invertebrates are widely used in environmental surveys (Dauvin et al., 2012). To facilitate the use of macro-invertebrates for biomonitoring, several biotic indices have recently been proposed, e.g. AMBI (Borja et al., 2000), BO2A (Dauvin and Ruellet, 2007), and BENTIX (Simboura and Zenetos, 2002).
Until now, few studies have been carried out to test and validate the performance of the widely used AMBI, BO2A, and BENTIX along the Brazilian coast (e.g. Brauko et al., 2016; Checon et al., 2018). These studies suggested that the inferred environmental quality based on these indices was, in general, satisfactory for the studied gradients in estuaries along the coasts of Brazil (Valença and Santos, 2012; Brauko et al., 2016; Checon et al., 2018; Souza et al., 2021b). However, some inconsistencies in the ecological classification provided by AMBI and other indices were reported (e.g. Brauko et al., 2016; Souza et al., 2021b). Specifically, a study in a subtropical estuary in Paranaguá Bay (Brazil) observed significant differences in the assignment of ecological quality status (EcoQS) among the tested indices (Brauko et al., 2016). The level of agreement in determining EcoQS varied depending on the index, and not all indices were accurately responsive to the varying levels of sewage contamination in the tidal flats (Brauko et al., 2016). This may be explained by the fact that the assignment of species to ecological groups (EGs) is based on taxa found in European waters (Borja et al., 2000; Simboura and Zenetos, 2002). Many species from Brazilian coastal and transitional ecosystems are not yet included in the database (Brauko et al., 2016; Muniz et al., 2005; Valença and Santos, 2012). Since these indices are based on the relative proportion of sensitive/tolerant taxa, assigning dominant species to one of the five EGs constitutes a prerequisite for the adequate evaluation of ecosystems, as Bigot et al. (2008) described. However, appropriate assignments are neither necessarily available (Muniz et al., 2005) nor easy to achieve beyond the coasts of Europe, for which the AMBI was developed (Borja et al., 2000). Along the Brazilian coasts, if macro-invertebrate taxa were used with the original European species list without modifying their EGs, most EcoQS scores would be wrongly evaluated (Valença and Santos, 2012). Hence, the applicability of these indices requires adjustments regarding some species' assignment in EGs (Checon et al., 2018; Muniz et al., 2005). Parallel to applying macro-invertebrate-based biotic indices to a broader extent, the complementary use of different indices and/or methods is recommended to confidently assess the EcoQS of coastal areas in Brazil (Muniz et al., 2005). Efforts should focus on developing integrative tools to help the environmental decision-making process (Muniz et al., 2011). For instance, benthic foraminifera are complementary to marine macro-invertebrates for characterising the environmental health of benthic ecosystems in Europe (Alve et al., 2019; Bouchet et al., 2020, 2018a, b).
Due to their wide distribution, small size, high abundance even in small amounts of sediment sample, short life cycles and reproductive cycles, high biodiversity, and specific ecological requirements (see Review in Schönfeld et al., 2012), foraminifera are particularly sensitive and can be successfully applied as bioindicators of environmental change in a wide range of transitional and marine habitats (e.g. Alve, 1995; Armynot du Châtelet et al., 2004; Francescangeli et al., 2020). There is a long-lasting tradition of ecological studies on benthic foraminifera (total assemblages or living fauna) in Brazil (e.g. Vilela et al., 2004; Burone et al., 2006; Debenay et al., 1998; Eichler et al., 2018; Eichler et al., 2003; Duleba et al., 2018). Specifically, living foraminiferal fauna have been utilised as sensitive indicators of trace metal pollution (e.g. Raposo et al., 2022), organic matter (OM) enrichment (e.g. Laut et al., 2016), oil spill (e.g. Nunes et al., 2023), shellfish farming (e.g. Rudorff et al., 2012), and water sewage (e.g. Filippos et al., 2023). To meet the objectives of the varying marine legislation, biotic indices have been established to synthesise the ecological information provided by foraminiferal communities (O'Brien et al., 2021). Some of these indices are based on diversity (Alve et al., 2009; Bouchet et al., 2012, 2018a), while others, such as Foram-AMBI, i.e. an adaptation of AMBI to benthic foraminifera (Alve et al., 2016; Bouchet et al., 2021; Jorissen et al., 2018), TSI-Med (Barras et al., 2014), and FSI (Dimiza et al., 2016), rely on the response of species along a pollution gradient, commonly ascribed to enrichment in total organic carbon (TOC). These indices have been applied to monitor the EcoQS in the context of aquaculture (e.g. Bouchet et al., 2020), trace metal elements, and water sewage (e.g. Melis et al., 2019). It is worth mentioning that lists of ecological behaviour of benthic foraminiferal species have already been compiled for some geographical areas and specific habitats, such as in the Northeast Atlantic and Arctic fjords on continental shelves and slopes (Alve et al., 2016); in open waters in the Mediterranean Sea (Jorissen et al., 2018); in transitional waters (TWs) along the English Channel and European coasts (Bouchet et al., 2021), called hereafter the European TW Atlantic list, and along the Mediterranean sea coasts (Bouchet et al., 2021), called hereafter the European TW Mediterranean list; and in the Gulf of Mexico (O'Malley et al., 2021). These species lists can be used in the different sensitivity-based indices designed for benthic foraminifera that consider the relative proportion of sensitive, tolerant, and opportunistic species to assess EcoQS, particularly Foram-AMBI. As for AMBI, there are no species lists adapted to Brazil, and adjusting local species classification in EGs to improve the accuracy of Foram-AMBI is required.
Preliminary tests of Foram-AMBI in Brazilian TWs are scarce (Nunes et al., 2023; Damasceno et al., 2024; Martins et al., 2020). For example, in Guanabara Bay, the increase in environmental stress was well correlated with Foram-AMBI (Nunes et al., 2023). The assessment of the EcoQS based on living benthic foraminifera illustrated the elevated pollution level in this bay (Cotovicz Jr. et al., 2015), showing the high potential of benthic foraminiferal biotic indices for the evaluation of environmental health (Nunes et al., 2023). However, the percentage of agreement in terms of EcoQS assessment between the diversity index exp() and Foram-AMBI was only ∼ 64 % (Nunes et al., 2023). The use of the European TW Atlantic list for computing Foram-AMBI may have limited the accuracy and reliability of the index in Brazilian waters, as observed for benthic macro-invertebrates (Muniz et al., 2005; Valença and Santos, 2012; Checon et al., 2018). According to Nunes et al. (2023), a better agreement between the indices can be attained with a specific Brazilian list of foraminiferal species' assignments to the EGs of Foram-AMBI. Overall, these results and previous studies are promising regarding the use of Foram-AMBI in TWs of Brazil; however, many local species remain unassigned or wrongly assigned to an EG. Indeed, geographical variability in benthic foraminiferal EG assignments has already been observed in Europe (Bouchet et al., 2021). Suitable application of Foram-AMBI demands further research on tolerance shifts of key indicator species in different geographical regions and habitats. This would significantly improve the EG assignment in the area, thereby increasing Foram-AMBI effectiveness and providing an important tool to monitor and preserve transitional ecosystems. Currently, only Norway recognises benthic foraminifera as a Biological Quality Element (BQE; Veileder, 2018). Although they have been widely used as accurate and reliable indicators of environmental conditions (O'Brien et al., 2021), they have not been included in the official Brazilian guidelines for environmental biomonitoring (Sousa et al., 2020).
In this context, this study aims to establish a foraminiferal species list adapted to TWs in Brazil to provide stakeholders with an improved and complementary method to benthic macro-invertebrates for environmental health assessment. To do that, we gathered and analysed published data from studies conducted in Brazilian TWs containing living foraminifera and environmental parameters. It was then possible to determine the indicative values of benthic foraminifera and to assign them to five EGs based on their responses to sediment TOC content, as described in the pioneering work on Foram-AMBI by Alve et al. (2016). The classification was tested by calculating Foram-AMBI with published foraminiferal data not used for the species assignment from two polluted transitional areas in Brazil, namely Sepetiba Bay (Damasceno et al., 2024) and Guanabara Bay (Nunes et al., 2023). We further compared species' assignments for those occurring in both Brazil and Europe to confirm any potential tolerance shift over a large geographical scale.
2.1 Dataset selection for the Brazilian list
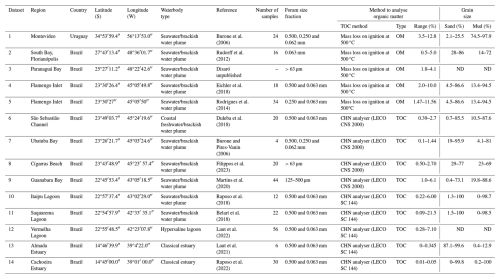
A literature collection was performed to find studies on living foraminifera and reporting relevant environmental parameters such as organic matter or total organic carbon content. We selected studies following the same criteria as those defined in previous work on the Foram-AMBI list (Alve et al., 2016; Bouchet et al., 2021; Jorissen et al., 2018). Only studies in coastal waters and TWs according to McLusky and Elliott (2007) were selected (Table 1). Furthermore, studies had to be solely based on living benthic foraminifera. Note that all datasets used in the present study are based on rose-bengal-stained fauna. The foraminiferal and TOC data had to be from the same station (less than 1 m from the sampling point). The TOC content data in some studies were derived from the measurement of loss on ignition (LOI; studies 1, 2, 3, 4, and 5 in Table A1). For these five studies, the TOC content was estimated by dividing by 3 the OM content obtained by LOI measurement as suggested for TW sediments from tropical areas (Leong and Tanner, 1999). Only samples containing at least 50 stained specimens were considered (Alve et al., 2016).
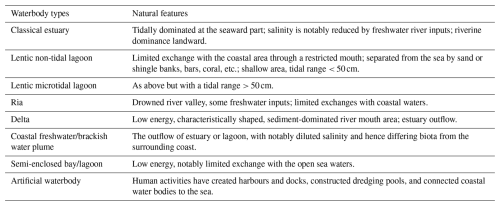
Table 1Waterbody types in intertidal areas and transitional waters, according to McLusky and Elliott (2007). The “artificial waterbody” type (European communities, 2000, p. 6) was added to complete the original table.
The selected studies were based on the > 63 or > 125 µm fraction (see Appendix A). In total, 14 studies were considered (Fig. 1, Table A1). Detailed information on the selected datasets can be found in Appendix A. We gathered all data in a master table (available in Bouchet et al., 2025) containing relative species abundances and TOC concentrations in sediments (in %) at each studied station selected. Species names were homogenised among studies following the World Register of Marine Species (WoRMS; Hayward et al., 2020a). Only accepted scientific names from WoRMS are used in this study (Hayward et al., 2020b), and the unique AphiaID is reported for each species.
2.2 Assignment to ecological groups
To assign foraminiferal species, we followed Bouchet et al. (2021) and used an objective method for species assignments in order to avoid using “best expert judgement”. The weighted-averaging (WA) optimum and tolerance (Birks et al., 1990; Ter Braak, 1987) were computed on the master table data (see Sect. 2.1 for data selection) for each species to determine its response to TOC (%). A simple and ecologically reasonable estimate of a benthic foraminiferal species' optimum is the average of all TOC values for intertidal areas and TWs in which the species occurs, weighted by the species' relative abundance (WA regression). To summarise, the WA optimum method is rapid, easy to implement, theoretically sound, and robust (Birks et al., 1990), and it leads to an objective assessment of species-specific indicative values.
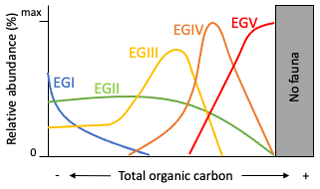
In detail, foraminiferal species were assigned to five EGs according to their response to organic carbon enrichment: (i) EGI – “sensitive species” are sensitive to TOC enrichment, and their relative abundance is highest at the lowest TOC values, dropping to zero as organic carbon concentration increases; (ii) EGII – “indifferent species” are unaffected by the organic carbon enrichment and never dominate the assemblage, occurring in low relative abundance over a broad range of organic carbon concentrations, only being absent at very high concentrations; (iii) EGIII – “tolerant species” are stimulated by the excess organic carbon enrichment, but they may occur at low TOC and are absent at very high organic carbon concentrations, being labelled as “third-order opportunistic species” by Jorissen et al. (2018); (iv) EGIV – “second-order opportunistic species” have a clear positive response to organic carbon enrichment with maximum abundances between the maxima of EGIII and EGV; and (v) EGV – “first-order opportunistic species” exhibit a clear positive response to excess organic carbon enrichment with maximum abundances at the higher level induced by organic load. A theoretical distribution of these EGs is represented in Fig. 2. Foraminifera are not able to survive extreme levels of TOC concentrations.
2.3 Datasets to test the Brazilian list
Published datasets from Sepetiba Bay (Damasceno et al., 2024) and Guanabara Bay (Nunes et al., 2023) were used to test the Brazilian list developed in the present work based on the datasets presented in Appendix A. Note that these two datasets from Sepetiba Bay and Guanabara Bay were not used for the species assignments to avoid any circular argument. For the analysis of living benthic foraminifera in Sepetiba Bay and Guanabara Bay, three replicates of sediment (from three independent box-corer deployments) were collected at each site and pooled together. Sediments were sieved on a 63 µm (Guanabara Bay) and 125 µm (Sepetiba Bay) fraction. Then, living foraminifera (rose-bengal-stained) were identified and counted.
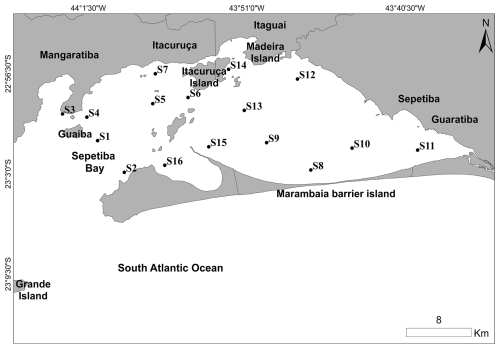
Sepetiba Bay is in the Rio de Janeiro State on the southeastern Atlantic coast of Brazil (Damasceno et al., 2024). The main opening, located between Ilha Grande and the tip of the Marambaia barrier island, connects the bay to the Atlantic Ocean. Sepetiba Bay is one of Brazil's most polluted coastal areas (Kütter et al., 2021). In the surroundings of the bay, there is the largest steelworks complex of Brazil; the highway known as the metropolitan arch of Rio de Janeiro State; the Santa Cruz air base; and three ports, including the harbour of Sepetiba/Itaguaí, which handles ≈ 51.7 million tonnes of iron ore per year. In addition, intense industrial discharge and domestic effluents occur in the area. Altogether, these anthropogenic activities have led to the contamination of the sediments and living organisms with potentially toxic elements (PTEs) (Ribeiro et al., 2015; Souza et al., 2021a). To investigate the foraminiferal response to environmental stress and to evaluate the EcoQS, 16 stations were sampled in Sepetiba Bay in May 2022 (Fig. 3). Environmental and foraminiferal data were published in Damasceno et al. (2024).
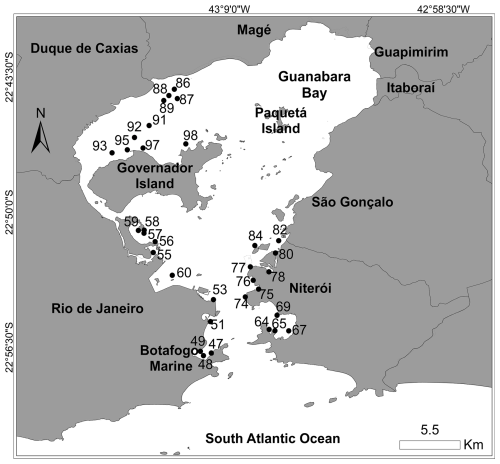
Guanabara Bay (Nunes et al., 2023) is located in the metropolitan region of Rio de Janeiro State (Fig. 4). Due to the massive presence of large urban centres and industries, the bay has experienced an enormous anthropogenic pressure (IBGE, 2014), which today represents a great environmental concern. The discharge of organic matter (OM), persistent organic pollutants, and heavy or trace metals has exerted significant pressures on this ecosystem (Baptista Neto et al., 2000, 2006, 2013). A total of 33 stations were sampled in the bay in the summer of 2018 (Fig. 4). Environmental and foraminiferal data were published by Nunes et al. (2023).
2.4 Calculation of Foram-AMBI
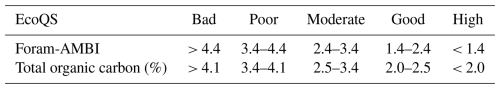
Foram-AMBI was applied to Sepetiba Bay and Guanabara Bay to evaluate the EcoQS. The foraminifera-specific criteria to assess EcoQS with Foram-AMBI are provided in Table 2 (Parent et al., 2021). If the count of living foraminifera assigned to EGs falls below 50 and/or the relative abundance of non-assigned species exceeds 20 %, the index was not calculated to prevent any biased estimation of the EcoQS (Borja and Muxika, 2005). The Foram-AMBI calculation follows the formula of Borja et al. (2000):
Table 2Criteria for determining the ecological quality status (EcoQS) according to Foram-AMBI (Parent et al., 2021) and with sediment TOC content (Bakke et al., 2010; Viaroli et al., 2004).
To evaluate the relevance of developing a regional species list, we computed Foram-AMBI on the Sepetiba Bay and Guanabara Bay datasets with the Brazilian and European TW Atlantic lists. We also computed Foram-AMBI with both lists on two European datasets: one from the French Atlantic coast in southwestern France in the Pertuis Charentais (Bouchet, 2007; Bouchet and Sauriau, 2008) and another from the harbour of Cagliari along the Sardinian coast in Italy (Schintu et al., 2016). The European Atlantic list, based on foraminiferal datasets from sites in the English Channel and other areas of the European Atlantic coast, reflects the ecology of species from the European TWs (Bouchet et al., 2021).
2.5 Statistical analyses
The weighted-averaging (WA) optimum and tolerance were computed for each species to determine its preference with respect to TOC (%) using the Analogue R package (Simpson and Oksanen, 2021). Since no criteria exist in the literature to infer EcoQS from TOC in Brazilian transitional areas, we used the ones from Bakke et al. (2010) and Viaroli et al. (2004) to assign foraminiferal species to EGs following the same procedure as in Bouchet et al. (2021). After optimum calculation with the WA method, species assignment to EGs was done as follows: if the species had an optimum in the TOC range of 0 %–2 %, it was assigned to EGI; in the range of 2 %–2.5 %, it was assigned to EGII; in the range of 2.5 %–3.4 %, it was assigned to EGIII; in the range of 3.4 %–4.1 %, it was assigned to EGIV; and above 4.1 %, it was assigned to EGV. This is based on TOC-derived EcoQS (see Table 2; Bakke et al., 2010; Viaroli et al., 2004). The outcome of this work is a list of species assigned to one of the EGs.
To further illustrate the classification, a typical example for each EG is shown to characterise species response along the TOC gradient. A locally weighted scatterplot smooth line (LOESS) was fitted to each scatterplot. Marginal plots were added to each scatterplot to show the frequency of distribution of occurrences along the TOC gradient. The median of the distribution of the occurrences was also computed.
We then compared the agreement between the EcoQS assessment with exp() (Nunes et al., 2023) and the one obtained with Foram-AMBI in Guanabara Bay in the present study. The contingency table was analysed using Cohen's kappa coefficient (Landis and Koch, 1977) from the package “irr” (Heinzl and Leisch, 2022). Two EcoQS categories (i.e. “acceptable” or “not acceptable”) were considered, where “acceptable” results from “High” or “Good” EcoQS and “not acceptable” results from “Moderate”, “Poor”, or “Bad” EcoQS (Blanchet et al., 2008; Bouchet and Sauriau, 2008). The following classification was used: (1) full agreement when both indices precisely identify the same EcoQS class, (2) partial agreement (i.e. different classes but the same category, namely acceptable and not acceptable), and (3) full disagreement when the two indices provide a different category in the EcoQS assessment.
Since data do not fit a normal distribution (Shapiro–Wilk test, p<0.05), the correlations between Foram-AMBI and environmental parameters were investigated using nonparametric Kendall's coefficient of rank correlation (τ). Kendall's coefficient of correlation was used instead of Spearman's coefficient of correlation (ρ) because Spearman's ρ provides greater weight to pairs of ranks that are further apart, while Kendall's τ weights each disagreement in rank equally (Sokal and Rohlf, 1995).
3.1 Foraminiferal species from southwestern Atlantic TW assignments to Foram-AMBI EGs
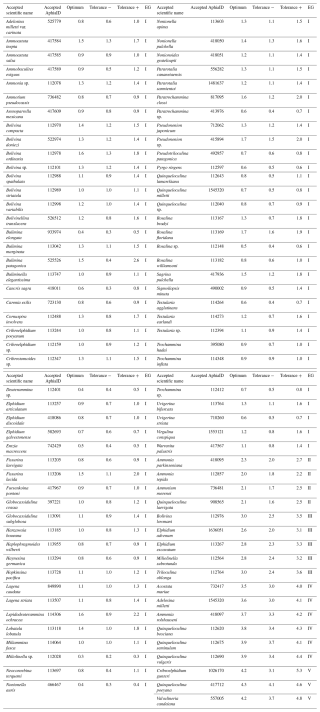
A total of 95 species from Brazilian and Uruguayan TWs were assigned: 77 in the sensitive EGI, 4 in the indifferent EGII, 5 in the tolerant EGIII, 6 in the second-order opportunistic EGIV, and 3 in the first-order opportunistic EGV (Fig. 5, Table B1). Figure 6 presents a typical example of species response curves for each of the five EGs, i.e. Buliminella elegantissima (EGI), Ammonia parkinsoniana (EGII), Ammonia tepida (EGIII), Quinqueloculina seminulum (EGIV), and Cribroelphidium gunteri (EGV).
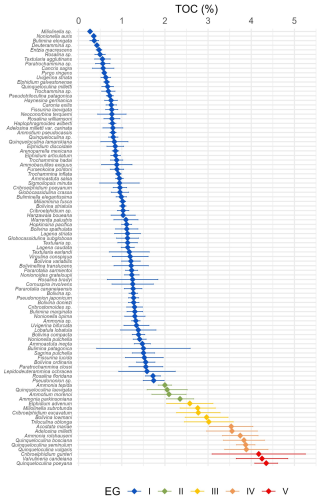
Figure 5Caterpillar plot of the Brazilian species list: species optima and tolerance interval against TOC and their assignment to EGs.
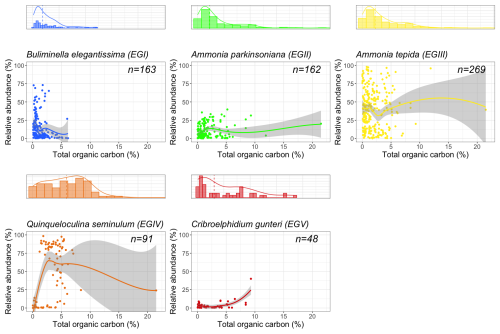
Figure 6Scatterplots fitted with a locally weighted scatterplot smooth line (LOESS) to visualise species response patterns along the TOC (%) gradient. Typical responses are given for each of the five Foram-AMBI EGs from southwestern Atlantic coastal waters and TWs (the shaded area is the 95 % confidence interval). Buliminella elegantissima (EGI), Ammonia parkinsoniana (EGII), Ammonia tepida (EGIII), Quinqueloculina seminulum (EGIV), and Cribroelphidium gunteri (EGV). Marginal plots show the frequency distribution of occurrences along the TOC gradient; the dashed line marks the median.
3.2 Test of the Brazilian TW list on independent datasets
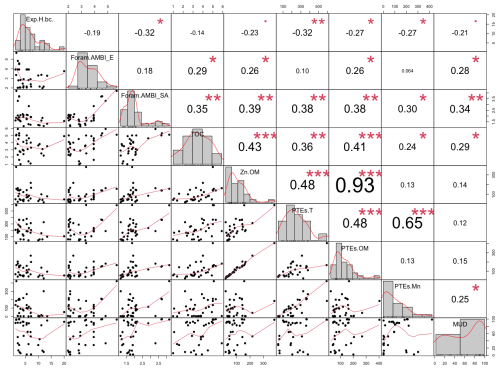
Based on the Brazilian list (Fig. 7a), stations at the entrance of Sepetiba Bay had a High/Good EcoQS (S01, S02, S05, and S06). Conversely, stations from the inner part of the bay were all in a Good EcoQS, except S13, which exhibited a High EcoQS. Foram-AMBI based on the Brazilian TW list significantly correlated with Cd (p<0.001), silt + clay, Hg, Pb, Zn (p<0.01), TOC, As, and Cr (p<0.05; Fig. C1).
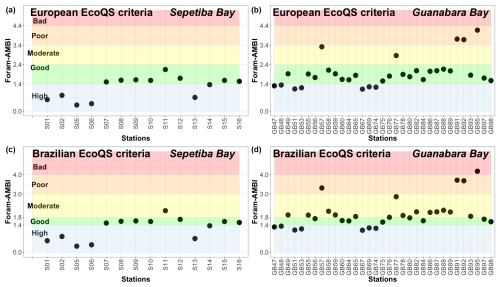
Figure 7Foram-AMBI calculated on independent datasets with the South American list and EcoQS using Parent et al. (2021) criteria for the (a) Sepetiba Bay (Damasceno et al., 2024) and (b) Guanabara Bay lists (Nunes et al., 2023) and EcoQS using criteria developed in the present study for Brazilian TWs for (c) Sepetiba Bay and (d) Guanabara Bay.
Based on the Brazilian list, 7 stations in Guanabara Bay had a High EcoQS, 21 had a Good EcoQS, 2 had a Moderate EcoQS, and 3 had a Poor EcoQS (Fig. 7b). Furthermore, Foram-AMBI computed with the Brazilian TW list significantly correlated with TOC, Zn in organic matter, total PTEs (PTEs.T), PTEs in organic matter (PTEs.OM), mud (p<0.01; Fig. C2), and PTEs in Mn (PTEs.Mn) (p<0.05; Fig. C2).
3.3 Comparison between Foram-AMBI and exp( in Guanabara Bay
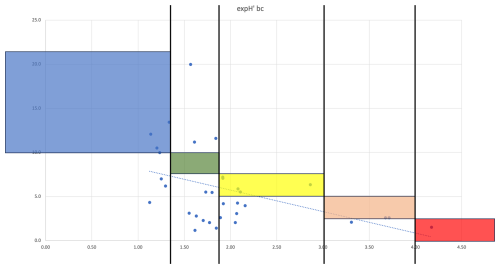
In Guanabara Bay, the Foram-AMBI values calculated with the Brazilian list were significantly correlated with exp() according to Kendall's coefficient (p<0.05; Fig. C2). However, the comparison of EcoQS values obtained in the present study – adopting Foram-AMBI criteria from Parent et al. (2021) – with those obtained with exp() in Nunes et al. (2023) showed a slight level of agreement of only about 36 %, with 18 % full agreement, 18 % partial agreement, and 64 % disagreement (Fig. 8a). The Cohen's Kappa results (p= 0.078) suggest that the low observed agreement is random.
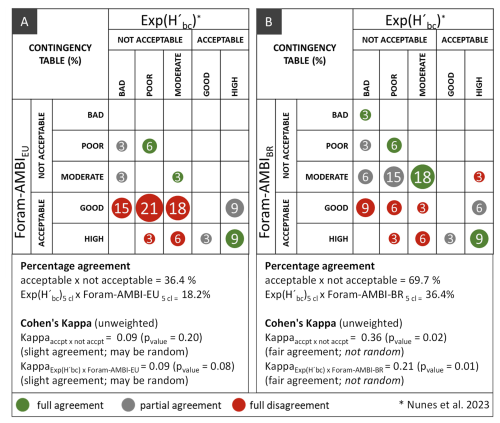
Figure 8The contingency table and Cohen's Kappa analysis results for the comparison of EcoQS obtained in Guanabara Bay with exp() and Foram-AMBI with the Brazilian list (the present work). (a) With Foram-AMBI criteria from Parent et al. (2021) and (b) with Foram-AMBI criteria established in the present work.
Given this rather low agreement, an attempt was made to intercalibrate EcoQS between exp() and Foram-AMBI to establish specific criteria for Brazilian TWs for the latter. A linear regression plot between the two indices was created, and, considering the good agreement of exp()'s EcoQS with the environmental conditions in Guanabara Bay (Nunes et al., 2023), limits between EcoQS for Foram-AMBI were calibrated according to those of exp() (Fig. D1). The new criteria are presented in Table 3, and the updated EcoQS is presented in Fig. 7c and d for Sepetiba Bay and Guanabara Bay, respectively.
Table 3New criteria for determining the ecological quality status (EcoQS) according to Foram-AMBI in Brazilian TWs.

According to the EcoQS established with the new Foram-AMBI criteria (Fig. 7c and d), a fair and statistically significant level of agreement of 70 % was obtained between Foram-AMBI and exp(), with 36 % full agreement, 33 % partial agreement, and 30 % disagreement (p<0.05; Fig. 8b). These results, based on the Brazilian TW list, were better than the ones obtained comparing exp() with Foram-AMBI calculated with the Atlantic TW European list, with a level of agreement of 64 %, with 15 % full agreement, 49 % partial agreement, and 36 % disagreement (Nunes et al., 2023).
3.4 Comparison between the Brazilian and the European Atlantic lists
The Brazilian list shares 29 species with the European TW Atlantic list. Except for 7 species (Ammonia parkinsoniana, A. tepida, Ammotium morenoi, Arenoparella mexicana, Cribroelphidium poeyanum, Elphidium advenum, and Lepidodeuterammina ochracea), assignments in EGs differ between the two lists (Table 4).
Table 4Comparison of EG assignments for species found in both the European TWs and the Brazilian TWs. NA: not assigned

Conversely to Foram-AMBI calculated with the Brazilian TW list, in Sepetiba Bay, Foram-AMBI was not correlated to any environmental parameter when using the European TW list (p>0.05; Fig. C1). Note that, with the European TW list, about 44 % of the stations had more than 20 % of species not assigned to an EG (only 12 % with the South American list). In Guanabara Bay, with the European Atlantic list, Foram-AMBI was only significantly correlated with TOC and Zn.OM, PTEs.OM, and mud (p<0.05; Fig. C2). Note that correlations were weaker with the European TW list than with the Brazilian one.
When applying the Brazilian list to foraminiferal datasets from European sites, we did not find any significant correlation between Foram-AMBI and TOC in the Pertuis Charentais (p>0.05) and in the harbour of Cagliari (p>0.05), while it was significantly correlated with the European Atlantic list in the Pertuis Charentais (p<0.05) and with the European Mediterranean list in Cagliari (p<0.01). Note that, in the harbour of Cagliari, about 62 % of the stations had more than 20 % of species not assigned to an EG when considering the Brazilian list.
4.1 Foram-AMBI performance in Brazilian transitional waters
A key prerequisite for effectively evaluating Brazilian coastal ecosystems with Foram-AMBI was the assignment of dominant species to one of the five EGs. This study reports species indicator values of 95 foraminiferal species from coastal waters and TWs from Brazil and, for the first time, assigns them to one of the five EGs used in Foram-AMBI. This work contributes to the ongoing work aimed at acknowledging benthic foraminifera as an official BQE (Alve et al., 2016). Furthermore, the Brazilian list complements the five existing lists: those established for the Northeast Atlantic and Arctic fjords, continental shelves, and slopes (Alve et al., 2016); for open environments in the Mediterranean Sea (Jorissen et al., 2018); for the English Channel and European Atlantic TWs (Bouchet et al., 2021); for the TWs in the Mediterranean Sea (Bouchet et al., 2021); and for the Gulf of Mexico (O'Malley et al., 2021).
The Brazilian list was tested on two independent datasets from Sepetiba Bay and Guanabara Bay to assess EcoQS with Foram-AMBI. Although we had significant correlations between Foram-AMBI and abiotic parameters, the EcoQS values were not in accordance with the known pollution in both bays. The criteria used for TOC and EcoQS were developed in and for TWs in Europe. Although recent works in Brazilian TWs have reported accurate biomonitoring with benthic foraminifera using these criteria (Nunes et al., 2023; Filippos et al., 2023), the use of local reference, hence local criteria, should be prioritised according to marine legislation. In Europe, the criteria to define EcoQS with the diversity index exp() were adapted to open environment (Bouchet et al., 2012) and TWs (Bouchet et al., 2018a), and the same was done for Foram-AMBI by calibrating the values used for macro-invertebrates to benthic foraminifera in open marine environments in the Mediterranean Sea (Parent et al., 2021). The discrepancies observed between EcoQS derived from Foram-AMBI with Parent et al. (2021) criteria and abiotic and between exp() and Foram-AMBI EcoQS assessment in the tested dataset from Guanabara Bay in terms of EcoQS actually highlight the fact that the criteria developed in Europe are not adapted for Brazilian TWs.
The EcoQS derived from Foram-AMBI with the new criteria developed in this study better reflect the high disturbances from human activities in these two ecosystems. There is a remarkable accumulation of metals in sediments in Sepetiba Bay (Ribeiro et al., 2015; Souza et al., 2021a), while Guanabara Bay exhibits a substantial anthropogenic impact from domestic sewage, industries, and oil spills (Baptista Neto et al., 2000, 2006, 2013). In Sepetiba Bay, the gradient from high to moderate EcoQS illustrates the intense pollution occurring in its inner part (Ribeiro et al., 2015; Damasceno et al., 2024). Furthermore, the moderate to poor EcoQS values in Guanabara Bay confirm the deleterious impact of pollution on the benthic habitats of this blue carbon ecosystem (Cotovicz et al., 2015). Significant correlations were also reported between Foram-AMBI and environmental parameters (e.g. TOC and metals). These results support the use of the list established in the present work and also confirm the reliability of Foram-AMBI for the evaluation of the environmental health in coastal and transitional areas in Brazil. The results of the tests showed that the WA optimum method allows an accurate assessment of the ecological requirement of each species along the TOC gradient as reported for European species from TWs (Bouchet et al., 2021). In Brazil, more tests on other independent datasets are, nevertheless, needed to further validate the findings of this work and most likely to improve the present study's list of species.
Furthermore, a significant correlation of Foram-AMBI with exp() in Guanabara Bay, together with congruent EcoQS, is reported. The exp() is known to be an accurate diversity index that can be used to monitor the health of benthic ecosystems in Brazil (Jesus et al., 2020; Filippos et al., 2023). However, in estuaries where diversity is naturally low, the use of exp() may lead to a wrong EcoQS assessment (Fouet et al., 2022). It may also happen in oligotrophic areas (Barras et al., 2014; Dubois et al., 2021). The difficulties of interpreting diversity indices and associated EcoQS values have also been discussed for macro-invertebrates (Labrune et al., 2006; Blanchet et al., 2008; Lavesque et al., 2009). This may be explained by the lack of ecological considerations in diversity indices (Bouchet and Sauriau, 2008). Specifically, exp() evaluates diversity, while Foram-AMBI is based on species tolerance level. In this context, and based on the present study results in Brazil, Foram-AMBI may serve, at a minimum, as a complementary index to exp() or even as an alternative when the use of exp() is not possible because of the natural environmental features of the studied ecosystems. Benthic foraminifera, as represented by the Foram-AMBI metric, may also complement benthic macro-invertebrates for a more exhaustive assessment of EcoQS, thereby contributing to the development of integrative tools to help the environmental decision-making process (Muniz et al., 2011). In estuaries where AMBI (macro-invertebrates) fails to evaluate EcoQS (Valença and Santos, 2012), Foram-AMBI (foraminifera) may actually be prioritised.
The intercalibration of EcoQS boundaries of Foram-AMBI for Brazilian TWs allows improved assessment of the health of benthic habitats in both bays. However, one may consider that these new criteria were obtained with a rather limited dataset that may not encompass all the natural variability in Brazilian TWs. Hence, future works should focus on testing these new criteria and their accuracy and reliability in other TWs in Brazil.
4.2 Regionally specific lists are recommended
In their paper devoted to macro-invertebrate indicator species, Zettler et al. (2013) stressed that “the use of fixed reference lists needs to be reconsidered, especially in areas with strong salinity gradients, like estuaries or the Baltic Sea, or eutrophic systems like Mediterranean lagoons”. Some species are, indeed, flexible enough to adapt to their environment. Consequently, they may change their autecology requirements along environmental gradients (see Review in Zettler et al., 2013, and references therein). Species even exhibit different responses to disturbance depending on their habitat and the source of disturbances; they behave as sensitive species in some environmental settings, while they can be tolerant or opportunistic somewhere else or when facing a different perturbation (Zettler et al., 2013). The use of a unique sensitivity/tolerance list for different geographical areas (such as in AMBI and comparable methods based on benthic macro-invertebrates) is therefore not recommended (Grémare et al., 2009), since it integrates the ecological requirements from species behaviour over a span of geographical regions and subregions that is too large and not on a local scale. Local adaptation of species' ecological requirements may hence lead to wrong ecological group assignments if only one global species list is used (Dauvin et al., 2010; Zettler et al., 2013). Similar features were recorded for benthic foraminifera (see Review in Alve, 1995).
In the present study, the same assignment in EGs is only obtained for 5 species among the 29 shared between the Brazilian and the European TW Atlantic lists. Similar discrepancies were also observed in Europe, where offsets of up to four categories in foraminiferal species assignments to EGs, depending on habitat type (different water bodies in coastal waters and TWs) or latitudinal gradient, were reported (northern Europe to the Mediterranean Sea; Bouchet et al., 2021). Noticeably, Haynesina germanica is a sensitive species in Brazil, while it was assigned to the tolerant group in Europe. This species, introduced in South America (Calvo-Marcilese and Langer, 2010), may be less adapted and therefore more sensitive in subtropical estuaries compared to European settings in its natural range of distribution. Conversely, Elphidium excavatum is tolerant in the Brazilian list and sensitive in Europe TWs. These incongruent assignments may be explained by (i) different populations with different ecological requirements for TOC content, suggesting plasticity in this species (see Discussion above), or by (ii) the occurrence of cryptic species. The presence of cryptic species in Elphidiidae is well documented (Darling and Wade, 2008), and we may hypothesise that E. excavatum from Brazil and Europe belongs to different phylotypes. Hence, the present findings can be the result of different cryptic lineages having different levels of tolerance to TOC. This hypothesis remains highly speculative, since the data used in this work are based on morphological identifications in the absence of any thorough molecular assessment of this species in Brazil and Europe. Phylogeographic studies may help to better distinguish and identify possible cryptic species to describe the distribution pattern of the different phylotypes. A combination of morphological and molecular taxonomy in large environmental surveys would clarify the ecological requirements of different cryptic lineages and help to assign appropriate indicator values to genetically distinct populations. Therefore, ecological studies on cryptic species in relation to the indicative value of species present a major topic for further scientific research projects. Indeed, a recent work on three phylotypes of Ammonia spp. in Europe suggested that they actually have different ecological requirements (Pavard et al., 2023).
The comparison between the Brazilian list (developed in this work) and the European Atlantic and Mediterranean TW lists (Bouchet et al., 2021) also provides compelling evidence that reliable ecological assessments can only be achieved when the local conditions are considered. In South America, the European Atlantic TW list was far less accurate than the Brazilian list to evaluate the EcoQS. In Europe, we have a significant correlation between Foram-AMBI and TOC only with the European TW lists and not with the Brazilian ones. The Brazilian list was particularly poorly adapted to most stations in the Mediterranean Sea in Europe, since more than 20 % of species were not assigned to an EG, leading to a non-robust and unreliable assessment of EcoQS (Borja and Muxika, 2005). The Mediterranean Sea encompasses completely different, mostly oligotrophic ecosystems, leading to different species responses to TOC compared to the more organic-rich TWs of the Atlantic and Brazilian coasts (Angel et al., 2000; Dubois et al., 2021; Hyams-Kazphan et al., 2009). By applying the European TW Atlantic list in Brazilian coastal habitats and the Brazilian list in European habitats, both lists show poor accuracy outside their biogeographic area. The same results were reported for benthic macro-invertebrates, and it was stressed that more studies are needed in Brazil to adjust local macro-invertebrate species classification in EGs to improve AMBI's performance (Checon et al., 2018; Muniz et al., 2005; Brauko et al., 2016). The present study, therefore, confirms the importance of validating EG classifications when transferring and applying them overseas, as previously observed for benthic macro-invertebrates (Aguado-Giménez et al., 2007; Borja and Muxika, 2005; Keeley et al., 2012). A joint research effort is therefore necessary in order to recognise regional differences in species pool and ecological tolerances.
4.3 Limitations of the study
Various methods were used in the different selected studies to measure organic matter, from the old-fashioned loss on ignition (LOI) up to the more sophisticated use of an elemental analyser (EA). While the estimation by the latter is more accurate, the LOI is still largely used in large surveys because it allows a quick and cheap measurement of OM in sediments (Luczak et al., 1997). It is well acknowledged that the LOI may overestimate OM content compared to the EA (Barillé-Boyer et al., 2003); hence, OM values had to be converted to TOC. As a result, the TOC and the OM data from the different studies may be compared with caution. Although it may have introduced a bias in our species assignment, the problem is probably fairly limited, as previously suggested (Bouchet et al., 2021). Indeed, the selected sites yield a wide range of environmental conditions, ensuring that our dataset is representative of the natural variability occurring in transitional waters along the Brazilian coasts. The inclusion of datasets from hypersaline lagoons such as Vermelha Lagoon (Laut et al., 2017, 2022) may be questioned, as they represent a peculiar type of transitional ecosystem. Noticeably, they present harsh conditions for benthic foraminifera due to elevated and highly variable salinity and temperature and frequent hypoxia in bottom waters (Stal, 2012). These systems commonly contain fine, organic-rich sediments that promote the buildup of toxic byproducts such as hydrogen sulfide (Stal, 2012). Limited water exchange further amplifies these stressors, creating a selective environment where only a few opportunistic or euryhaline species can persist. Future works may explore if specific assignments should take place for foraminiferal species thriving in these peculiar lagoons.
The definition of a foraminiferal species list for the use of biotic indices in biomonitoring is based on the paradigm that TOC is one of the main constraining drivers of species distribution patterns in TWs (see Review in O'Brien et al., 2021). A further environmental parameter constraining foraminiferal species distribution is sediment grain size. In TWs, silt, clay, and OM, sedimentary contents are naturally high. The complex interplay between sediment grain size and TOC with foraminiferal species is complex. In their work in marginal environments from the Skagerrak and the Kattegat, Alve and Murray (1999) did not observe any clear trend in foraminiferal assemblage distribution patterns, TOC, and grain size. However, in the Canche estuary in the English Channel, both abiotic factors constrained foraminiferal assemblages, with sediment grain size being the limiting factor (Armynot du Châtelet et al., 2009). The fact that TOC in TWs may reach very high values, somewhat higher than in polluted environments, is due purely to the development of healthy natural vegetation and associated fauna developing within that environment (Armynot du Châtelet et al., 2018). Commonly, OM is more lignified and not labile (Armynot du Châtelet et al., 2009) and may not be regarded as a stress for benthic foraminifera, explaining why sediment grain size is usually the most limiting factor. In fact, TOC only informs about the reservoir of organic carbon, but it does not give any information about the quality and the origin of organic matter (Pusceddu et al., 2003). Sediment grain size also reflects the hydrodynamic regime and is linked with variations in associated environmental parameters, such as nutrients (OM quality), pollutant accumulation, oxygen, and active biogeochemical reactions on the sea floor, to which foraminifera react (Martins et al., 2015). Hence, several natural stressors must be considered when evaluating species' response to TOC. Noticeably, a species' tolerance to a stressor or a cocktail of stressors of human origin depends on whether the prevailing natural environmental conditions fall well within its ecological requirements or lie near the margins of its ecological niche (Alve, 1995). A taxon may exhibit greater resilience on anthropogenic stressors when the surrounding environment is close to its optimal conditions, compared to situations where it is already near the limits of its natural distribution. To summarise, the natural environmental features of a studied site may be just as important as the type of pollution in determining which sensitive, tolerant, or opportunistic species dominate in the foraminiferal assemblage (Alve, 1995).
In the present study, different size fractions are used to monitor EcoQS in Sepetiba Bay and Guanabara Bay, i.e. > 125 µm in the former and > 63 µm in the latter. In Norwegian fjords, a recent study tested the effect of the studied foraminiferal size fraction > 63 µm versus > 125 µm on the EcoQS assignment with different biotic indices, i.e. Shannon index, Hurlbert rarefaction index, Foram-AMBI, and Norwegian Quality Index (NQI; Klootwijk and Alve, 2022). The different size fractions had similar EcoQS values; hence they tended not to influence the Foram-AMBI results, for instance. Although we did not test it in Brazilian transitional ecosystems, we assume from the results of the Norwegian example that the different size fractions in Sepetiba Bay and Guanabara Bay did not introduce a bias in our conclusions. This should, however, be further tested in future works.
Last but not least, it is true that different taxonomical schools co-exist in Brazil, like everywhere else. This may hence lead to some inconsistencies between research groups in terms of species identification. Previous studies stressed this issue by assigning benthic foraminifera in EGs (Alve et al., 2016; Jorissen et al., 2018; Bouchet et al., 2021). Hence, there is an urgent need to start organising taxonomical workshops to intercalibrate the different taxonomical schools. For instance, the French researchers recently met for a taxonomical workshop, and, using morphological and molecular assessment, they homogenised their taxonomy on benthic foraminifera from French TWs. The outcome of this effort is a taxonomical guide (Jorissen et al., 2023) which serves as a reference at the French level and could serve as a basis to homogenise the taxonomy of foraminiferal species at Brazilian and international scales.
This work represents the first attempt to assign foraminiferal species to EGs from the southwestern Atlantic transitional waters and to investigate the potential applicability of Foram-AMBI using a regional list. This list is a key step towards improving biomonitoring surveys conducted in Brazil with benthic foraminifera. This may contribute to this group being regarded as an official BQE by the National Environment Council in Brazil. In this study, we reveal differences in the classification of EGs between species occurring in Brazil and in Europe. The overall pattern of EcoQS at the sites analysed would be different depending on the assignment of the selected species/taxa. For instance, if these taxa were submitted to the European list without modification of their EG, most sites would result in a different EcoQS. Hence, the outcome of the present work strongly supports the rationale for (i) developing regional lists for Foram-AMBI and (ii) establishing local criteria for EcoQS assessment. Our results further show that Foram-AMBI is robust in detecting the effects of different contaminants in the area and reinforce the importance of the index as a tool for coastal management.
To summarise, these results point in a promising direction. However, despite the encouraging findings of this study, they do not yet justify the straightforward implementation of Foram-AMBI in Brazilian TWs. To thoroughly assess the effectiveness of Foram-AMBI, we require further studies across a broader range of pollution gradients and to thoroughly consider the natural variability in Brazilian TWs that lie from the equatorial/tropical to the early temperate zones of the Southern Hemisphere. As in the case of earlier studies assigning benthic foraminifera to EGs in Europe, we anticipate that future applications of the Brazilian lists on SW Atlantic coasts will lead to regular updates. This would greatly improve the ecological group classifications in the area, thereby increasing Foram-AMBI effectiveness and providing an important tool to monitor and preserve Brazilian coastal ecosystems.
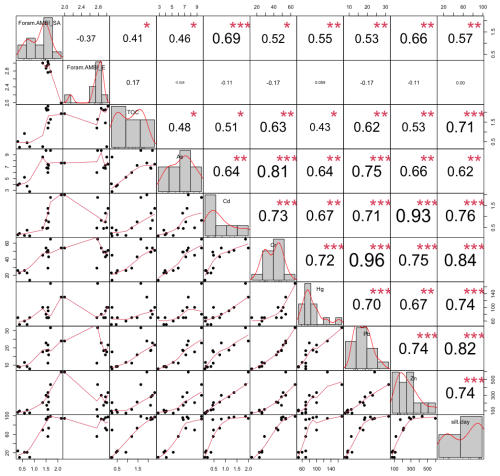
Figure C1Kendall's coefficient of rank correlation (τ) between environmental parameters (from Damasceno et al., 2024) and Foram-AMBI calculated with the Brazilian TW list (Foram.AMBI_SA) and the European TW list (Foram.AMBI_E) in Sepetiba Bay.
The data are available in Mendeley: https://doi.org/10.17632/nw4xd5ymgd.1 (Bouchet et al., 2025).
VMPB: conceptualisation, formal analysis, funding acquisition, methodology, visualisation, writing (original draft). SHdMS: conceptualisation, data curation, investigation, writing (original draft). PB: investigation, writing (review and editing). CB: investigation, formal analysis, visualisation, writing (review and editing). LB: investigation, writing (review and editing). WD: investigation, writing (review and editing). FFra: formal analysis, writing (review and editing). FFro: conceptualisation, data curation, methodology, writing (original draft). LL: investigation, writing (review and editing). DSP: investigation, writing (review and editing). ARR: investigation, writing (review and editing). STD: investigation, writing (review and editing). DVP: investigation, writing (review and editing). FLD: investigation, visualisation, writing (review and editing). JCP: data curation, writing (review and editing). MVAM: conceptualisation, data curation, investigation, writing (original draft).
The contact author has declared that none of the authors has any competing interests.
Publisher' note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. While Copernicus Publications makes every effort to include appropriate place names, the final responsibility lies with the authors.
This article is part of the special issue “Advances and challenges in modern and benthic foraminifera research: a special issue dedicated to Professor John Murray”. It is not associated with a conference.
The authors would like to thank the UMR8187 Laboratoire d’Océanologie et de Geosciences for funding the travel to Wimereux Marine Station, France, of Maria Virginia Alves Martins, Silvia Helena de Mello e Sousa, Fabrizio Frontalini, Sibelle Trevisan Disaró, and Fabio Francescangeli via the IRD support to initiate the work presented here. This work benefited from the technical support of the EMIMA experimental platform from the University of Lille. The authors thank the handling editor Andy Gooday and Michael Martínez-Colón, Luisa Bergamin, and one anonymous reviewer for their valuable comments and suggestions.
This research has been supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (grant nos. 311801/2021–8 to Silvia Sousa; 409479/2022-5 and 305001/2023-0 to Lazaro Laut; and 443662/2018-5, 302676/2019-8, and 305719/2023-8 to Virginia Martins), the Fundação de Amparo à Pesquisa do Estado de São Paulo (grant nos. 08/50938-7 and 09/51031-8 to Wania Duleba), the Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (grant nos. E-26/204.093/2022 to Fabricio Damasceno and E-26/211.278/2021, E-26/202.927/2019, and E-26/200.333/2023 to Virginia Martins), the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (grant no. 88881.982468/2024-01 to Fabricio Damasceno), and the Institut de Recherche pour le Développement (grant to Vincent Bouchet).
The publication of this article was funded by the Swedish Research Council, Forte, Formas, and Vinnova.
This paper was edited by Andy Gooday and reviewed by Luisa Bergamin, Michael Martínez-Colón, and one anonymous referee.
Aguado-Giménez, F., Marin, A., Montoya, S., Marin-Guirao, L., Piedecausa, A., and Garcia-Garcia, B.: Comparison between some procedures for monitoring offshore cage cultures in western Mediterranean Sea: sampling methods and impact indicators in soft substrata, Aquaculture, 271, 357–370, 2007.
Alve, E.: Benthic foraminiferal responses to estuarine pollution: a review, J. Foramin. Res., 25, 190–203, 1995.
Alve, E. and Murray, J. W.: Marginal marine environments of the Skagerrak and Kattegat: a baseline study of living (stained) benthic foraminiferal ecology, Palaeogeogr. Palaeocl., 146, 171–193, 1999.
Alve, E., Lepland, A., Magnusson, J., and Backer-Owe, K.: Monitoring strategies for re-establishment of ecological reference conditions: Possibilities and limitations, Mar. Pollut. Bull., 59, 297–310, https://doi.org/10.1016/j.marpolbul.2009.08.011, 2009.
Alve, E., Korsun, S., Schönfeld, J., Dijkstra, N., Golikova, E., Hess, C., Husum, K., and Panieri, G.: Foram-AMBI: A sensitivity index based on benthic foraminiferal faunas from North-East Atlantic and Arctic fjords, continental shelves and slopes, Mar. Micropaleontol., 122, 1–12, https://doi.org/10.1016/j.marmicro.2015.11.001, 2016.
Alve, E., Hess, S., Bouchet, V. M. P., Dolven, J. K., and Rygg, B.: Intercalibration of benthic foraminiferal and macrofaunal biotic indices: an example from the Norwegian Skagerrak coast (NE North Sea), Ecol. Indic., 96, 107–115, 2019.
Angel, D. L., Verghese, S., Lee, J. J., Saleh, A. M., Zuber, D., Lindell, D., and Symons, A.: Impact of a net cage fish farm on the distribution of benthic foraminifera in the northern Gulf of Eilat (Aqaba, Red Sea), J. Foramin. Res., 30, 54–65, 2000.
Armynot du Châtelet, E., Debenay, J.-P., and Soulard, R.: Foraminiferal proxies for pollution monitoring in moderately polluted harbors, Environ. Pollut., 127, 27–40, https://doi.org/10.1016/s0269-7491(03)00256-2, 2004.
Bakke, T., Kalqvist, T., Ruus, A., Breedveld, G. D., and Hylland, K.: Development of sediment quality criteria in Norway, J. Soil. Sediment., 10, 172–178, 2010.
Baptista Neto, J. A., Smith, B. J., and McAlister, J. J.: Heavy metal concentration in surface sediments in a nearshore environment, Jurujuba Sound, Southeast Brazil, Environ. Pollut., 109, 1–9, 2000.
Baptista Neto, J. A., Gingele, F. X., Leipe, T., and Brehme, I.: Spatial distribution of heavy metals in surficial sediments from Guanabara Bay: Rio de Janeiro, Brazil, Environ. Geol., 49, 1051–1063, https://doi.org/10.1007/s00254-005-0149-1, 2006.
Baptista Neto, J. A., Barreto, C. F., Silva, M. A. M., Smith, B. J., Mcallister, J. J., and Vilela, C. G.: Nearshore sedimentation as a record of land use change and erosion: Jurujuba Sound, Niterói, SE Brazil, Ocean Coast Manage., 77, 31–39, https://doi.org/10.1016/j.ocecoaman.2012.04.022, 2013.
Barillé-Boyer, A.-L., Barillé, L., Masse, H., Razet, D., and Heral, M.: Correction for particulate organic matter as estimated by loss on ignition in estuarine ecosystems, Estuar. Coast. Shelf S., 58, 147–153, 2003.
Barras, C., Jorissen, F. J., Labrune, C., Andral, B., and Boissery, P.: Live benthic foraminiferal faunas from the French mediterranean coast: towards a new biotic index of environmental quality, Ecol. Indic., 36, 719–743, 2014.
Belart, P., Clemente, I., Raposo, D., Habib, R., Volino, E., Villar, A., Alves Martins, M., Fontana, L., Lorini, M., Panigai, G., Frontalini, F., Figueiredo, M., Vasconcelos, S., and Laut, L.: Living and dead Foraminifera as bioindicators in Saquarema Lagoon System, Brazil, Lat. Am. J. Aquat. Res., 46, 1055–1072, https://doi.org/10.3856/vol46-issue5-fulltext-18, 2018.
Bigot, L., Grémare, A., Amouroux, J. M., Frouin, P., Maire, O., and Gaertner, J. C.: Assessment of the ecological quality status of soft-bottoms in Reunion Island (tropical Southwest Indian Ocean) using AZTI marine biotic indices, Mar. Pollut. Bull., 56, 704–722, 2008.
Birks, H. J. B., Line, J. M., Juggins, S., Stevenson, A. C., and Ter Braak, C. J. F.: Diatoms and pH reconstruction, Phil. T. R. Soc. Lon. B, 327, 263–278, 1990.
Blanchet, H., Lavesque, N., Ruellet, T., Dauvin, J.-C., Sauriau, P. G., Desroy, N., Desclaux, C., Leconte, M., Bachelet, G., Janson, A.-L., Bessineton, C., Duhamel, S., Jourde, J., Mayot, S., Simon, S., and de Montaudouin, X.: Use of biotic indices in semi-enclosed coastal ecosystems and transitional waters habitats – implications for the implementation of the European water framework directive, Ecol. Indic., 8, 360–372, 2008.
Borja, A. and Muxika, I.: Guidelines for the use of AMBI (AZTI's Marine Biotic Index) in the assessment of the benthic ecological quality, Mar. Pollut. Bull., 50, 787–789, 2005.
Borja, A., Franco, J., and Perez, V.: A marine biotic index to establish the ecological quality of soft-bottom benthos within European estuarine and coastal environments, Mar. Pollut. Bull., 40, 1100–1114, 2000.
Bouchet, V. M. P.: Dynamique et réponse fonctionnelle des foraminifères et de la macrofaune benthiques en zone ostréicole dans les pertuis charentais, Thèse de doctorat, University of Angers, 404 pp., http://www.theses.fr/2007ANGE0045 (last access: 14 August 2025), 2007.
Bouchet, V. M. P. and Sauriau, P.-G.: Influence of oyster culture practices and environmental conditions on the ecological quality of intertidal mudflats in the Pertuis Charentais (SW France): a multi-index approach, Mar. Pollut. Bull., 56, 1898–1912, 2008.
Bouchet, V. M. P., Goberville, E., and Frontalini, F.: Benthic foraminifera to assess the Ecological Quality Status of Italian transitional waters, Ecol. Indic., 84, 130–139, 2018a.
Bouchet, V. M. P., Telford, R. J., Rygg, B., Oug, E., and Alve, E.: Can benthic foraminifera serve as proxies for changes in benthic macrofaunal community structure? Implications for the definition of reference conditions, Mar. Environ. Res., 137, 24–36, 2018b.
Bouchet, V. M. P., Alve, E., Rygg, B., and Telford, R. J.: Benthic foraminifera provide a promising tool for ecological quality assessment of marine waters, Ecol. Indic., 23, 66–75, 2012.
Bouchet, V. M. P., Deldicq, N., Baux, N., Dauvin, J.-C., Pezy, J.-P., Seuront, L., and Méar, Y.: Benthic foraminifera to assess ecological quality statuses: the case of salmon fish farming, Ecol. Indic., 117, 106607, https://doi.org/10.1016/j.ecolind.2020.106607, 2020.
Bouchet, V. M. P., Frontalini, F., Francescangeli, F., Sauriau, P.-G., Geslin, E., Martins, M. V. A., Almogi-Labin, A., Avnaim-Katav, S., Di Bella, L., Cearreta, A., Coccioni, R., Costelloe, A., Dimiza, M. D., Ferraro, L., Haynert, K., Martinez-Colon, M., Melis, R., Schweizer, M., Triantaphyllou, M., Tsujimoto, A., Wilson, B., and Armynot du Châtelet, E.: Indicative value of benthic foraminifera for biomonitoring: assignment to ecological groups of sensibility to total organic carbon of species from European intertidal areas and transitional waters, Mar. Pollut. Bull., 164, 112071, https://doi.org/10.1016/j.marpolbul.2021.112071, 2021.
Bouchet, V. M. P., Sousa, S. H. M., Bonetti, C., Burone, L., Belart, P., Duleba, W., Francescangeli, F., Frontalini, F., Laut, L., Raposo, D., Rodrigues, A., Disaró, S. T., Pupo, D., Damasceno, F., Pavard, J.-C., Martins, M. V. A.: Living foraminifera relative abundances and total organic carbon in Brazilian transitional waters, Mendeley Data [data set], https://doi.org/10.17632/nw4xd5ymgd.1, 2025.
Brauko, K. M., Muniz, P., de Castro Martins, C., and da Cunha Lana, P.: Assessing the suitability of five benthic indices for environmental health assessment in a large subtropical South American estuary, Ecol. Indic., 64, 258–265, 2016.
Brazil: Lei no 6.938, de 31 de agosto de 1981. Dispõe sobre a Política Nacional do Meio Ambiente, seus fins e mecanismos de formulação e aplicação, e dá outras providências. Diário Oficial da União, Brasília, DF: Imprensa Oficial, http://www.planalto.gov.br/ccivil_03/leis/l6938.htm (last access: 14 August 2025), 1981.
Brazil: Conselho Nacional de Meio Ambiente. Resolução CON- AMA no 357, de 17 de março de 2005. Dispõe sobre a classificação dos corpos de água e diretrizes ambientais para o seu enquadramento, bem como estabelece as condições e padrões de lançamento de efluentes, e dá outras providências. Diário Oficial da União, Brasília, DF: Imprensa Oficial, https://conama.mma.gov.br/index.php?id=15876 (last access: 14 August 2025), 2005.
Burone, L. and Pires-Vanin, A. M. S.: Foraminiferal assemblages in Ubatuba Bay, south-eastern Brazilian coast, Sci. Mar., 70, 203–217, 2006.
Burone, L., Venturini, N., Sprechmann, P., Valente, P., and Muniz, P.: Foraminiferal responses to polluted sediments in the Montevideo coastal zone, Uruguay, Mar. Pollut. Bull., 52, 61–73, https://doi.org/10.1016/j.marpolbul.2005.08.007, 2006.
Calvo-Marcilese, L. and Langer, M. R.: Breaching biogeographic barriers: the invasion of Haynesina germanica (Foraminifera, Protista) in the Bahía Blanca estuary, Argentina, Biol. Invasions, 12, 3299–3306, 2010.
Checon, H. H., Corte, G. N., Muniz, P., Brauko, K. M., Di Domenico, M., Bícego, M. C., Siegle, E., Figueira, R. C. L., and Amaral, A. C. Z.: Unraveling the performance of the benthic index AMBI in a subtropical bay: The effects of data transformations and exclusion of low-reliability sites, Mar. Pollut. Bull., 126, 438–448, 2018.
Cotovicz Jr., L. C., Knoppers, B. A., Brandini, N., Costa Santos, S. J., and Abril, G.: A strong CO2 sink enhanced by eutrophication in a tropical coastal embayment (Guanabara Bay, Rio de Janeiro, Brazil), Biogeosciences, 12, 6125–6146, https://doi.org/10.5194/bg-12-6125-2015, 2015.
Damasceno, F. L., Martins, M. V. A., Frontalini, F., Pawlowski, J., Cermakova, K., Angeles, I. B., Costa Santos, L. G., Filho, J. G. M., Francescangeli, F., Senez-Mello, T. M., Castelo, W. F. L., Rebouças, R. C., Duleba, W., Sousa, S. H. M., Laut, L., and Antonioli, L.: Assessment of the ecological quality status of the Sepetiba Bay (SE Brazil): When metabarcoding meets morphology on foraminifera, Mar. Environ. Res., 195, 106340, https://doi.org/10.1016/j.marenvres.2024.106340, 2024.
Darling, K. F. and Wade, C. M.: The genetic diversity of planktic foraminifera and the global distribution of ribosomal RNA genotypes, Mar. Micropaleontol., 67, 216–238, 2008.
Dauvin, J.-C. and Ruellet, T.: Polychaete/amphipod ratio revisited, Mar. Pollut. Bull., 55, 215–224, 2007.
Dauvin, J.-C., Bellan, G., and Bellan-Santini, D.: Benthic indicators: From subjectivity to objectivity – Where is the line?, Mar. Pollut. Bull., 60, 947–953, 2010.
Dauvin, J. C., Alizier, S., Rolet, C., Bakalem, A., Bellan, G., Gesteira, J. G., Grimes, S., De-La-Ossa-Carretero, J., and Del-Pilar-Ruso, Y.: Response of different benthic indices to diverse human pressures, Ecol. Indic., 12, 143–153, 2012.
Debenay, J.-P., Eichler, B. B., Duleba, W., Bonetti, C., and Eichler-Coelho, P.: Water stratification in coastal lagoons: its influence on foraminiferal assemblages in two Brazilian lagoons., Mar. Micropaleontol., 35, 65–89, 1998.
Diaz, R. and Rosenberg, R.: Spreading dead zones and consequences for marine ecosystems, Science, 321, 926–929, 2008.
Dimiza, M. D., Triantaphyllou, M. V., Koukousioura, O., Hallock, P., Simboura, N., Karageorgis, A. P., and Papathanassiou, E.: The Foram Stress Index: A new tool for environmental assessment of soft-bottom environments using benthic foraminifera. A case study from the Saronikos Gulf, Greece, Eastern Mediterranean, Ecol. Indic., 60, 611–621, 2016.
Dubois, A., Barras, C., Pavard, J.-C., Donnay, A., Beatrix, M., and Bouchet, V. M. P.: Distribution patterns of benthic foraminifera in fish farming areas (Corsica, France): implications for the implementation of biotic indices in biomonitoring studies, Water, 13, 2821, https://doi.org/10.3390/w13202821, 2021.
Duleba, W., Teodoro, A. C., Debenay, J.-P., Alves Martins, M. V., Gubitoso, S., Pregnolato, L. A., Lerena, L. M., Prada, S. M., and Bevilacqua, J. E.: Environmental impact of the largest petroleum terminal in SE Brazil: a multiproxy analysis based on sediment geochemistry and living benthic foraminifera, PLoS One, 13, e0191446, https://doi.org/10.1371/journal.pone.0191446, 2018.
Eichler, P., McGann, M., Rodrigues, A. R., Mendonça, A., Amorim, A., Bonetti, C., Cordeiro de Farias, C., Mello e Sousa, S. H., Vital, H., and Praxedes Gome, M.: The occurrence of the invasive foraminifera Trochammina hadai Uchio in Flamengo Inlet, Ubatuba, São Paulo State, Brazil, Micropalaeontology, 64, 391–402, 2018.
Eichler, P. P. B., Eichler, B. B., de Miranda, L. B., Pereira, E. R. M., Kfouri, P. B. P., Pimenta, F. M., Bérgamo, A. L., and Vilela, C. G.: Benthic foraminiferal response to variations in temperature, salinity, dissolved oxygen and organic carbon. the Guanabara Bay, Rio de Janeiro, Brazil, Anuario do Instituto de Geociencias, 26, 36–51, 2003.
European Communities: Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for community action in the field of water policy, Off. J. Eur. Communities, 43, 75, https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:02000L0060-20140101 (last access: 14 August 2025), 2000.
Filippos, L. S., Duleba, W., Hohenegger, J., Pregnolato, L. A., Bouchet, V. M. P., and Martins, M. V. A.: Domestic Sewage Outfall Severely Altered Environmental Conditions, Foraminiferal Communities, and Ecological Quality Statuses in Front of the Nearshore Beach of Cigarras (SE Brazil), Water, 15, 405, https://doi.org/10.3390/w15030405, 2023.
Fouet, M. P. A., Singer, D., Coynel, A., Héliot, S., Howa, H., Lalande, J., Mouret, A., Schweizer, M., Tcherkez, G., and Jorissen, F. J.: Foraminiferal Distribution in Two Estuarine Intertidal Mudflats of the French Atlantic Coast: Testing the Marine Influence Index, Water, 14, 645, https://doi.org/10.3390/w14040645, 2022.
Francescangeli, F., Quijada, M., Armynot du Châtelet, E., Frontalini, F., Trentesaux, A., Billon, G., and Bouchet, V. M. P.: Multidisciplinary study to monitor consequences of pollution on intertidal benthic ecosystems (Hauts de France, English Channel, France): comparison with natural areas, Mar. Environ. Res., 160, 105034, https://doi.org/10.1016/j.marenvres.2020.105034, 2020.
Grémare, A., Labrune, C., Vanden Berghe, E., Amouroux, J. M., Bachelet, G., Zettler, M. L., Vanaverbeke, J., Fleischer, D., Bigot, L., Maire, O., Deflandre, B., Craeymeersch, J., Degraer, S., Dounas, C., Duineveld, G., Heip, C., Herrmann, M., Hummel, H., Karakassis, I., Kedra, M., Kendall, M., Kingston, P., Laudien, J., Occhipinti-Ambrogi, A., Rachor, E., Sardá, R., Speybroeck, J., Van Hoey, G., Vincx, M., Whomersley, P., Willems, W., Wlodarska-Kowalczuk, M., and Zenetos, A.: Comparison of the performances of two biotic indices based on the MacroBen database, Mar. Ecol. Prog. Ser., 382, 297–311, 2009.
Hayward, B. W., Le Coze, F., Vachard, D., and Gross, O.: World Foraminifera Database, WORMS [data set], https://doi.org/10.14284/305, 2020b.
Hayward, B. W., Le Coze, F., Vandepitte, L., and Vanhoorne, B.: Foraminifera in the World Register of Marine Species (WoRMS) taxonomic database, J. Foramin. Res., 50, 291–300, https://doi.org/10.2113/gsjfr.50.3.291, 2020a.
Heinzl, H. and Leisch, F.: irr: Various Coefficients of Interrater Reliability and Agreement, R package version 0.84.1 [code], https://CRAN.R-project.org/package=irr (last access: 14 August 2025), 2022.
Hyams-Kazphan, O., Almogi-Labin, A., Benjamini, C., and Herut, B.: Natural oligotrophy vs. pollution-induced eutrophy on the SE Mediterranean shallow shelf (Israel): environmental parameters and benthic foraminifera, Mar. Pollut. Bull., 58, 1888–1902, 2009.
IBGE: Rio de Janeiro, in: Anuario Estatístico Do Brasil, vol. 74, https://biblioteca.ibge.gov.br/visualizacao/periodicos/20/aeb_2014.pdf (last access: 13 February 2022), 2014.
Jesus, S. d. S. M., Frontalini, F., Bouchet, V. M. P., Yamashita, C., Sartoretto, J. R., Figueira, R. C. L., and Sousa, S. H. M.: Reconstruction of paleo-ecological quality statuses in an anthropogenised estuary using benthic foraminifera: the Santos estuary (São Paulo State, SE Brazil), Mar. Environ. Res., 162, 105121, https://doi.org/10.1016/j.marenvres.2020.105121, 2020.
Jorissen, F. J., Nardelli, M. P., Almogi-Labin, A., Barras, C., Bergamin, L., Bicchi, E., El Kateb, A., Ferraro, L., McGann, M., Morigi, C., Romano, E., Sabbatini, A., Schweizer, M., and Spezzaferri, S.: Developing Foram-AMBI for biomonitoring in the Mediterranean: Species assignments to ecological categories, Mar. Micropaleontol., 140, 33–45, 2018.
Jorissen, F., Fouet, M., Armynot du Châtelet, É., Barras, C., Bouchet, V., Daviray, M., Francescangeli, F., Geslin, E., Le Moigne, D., Licari, L., Mojtahid, M., Nardelli, M.-P., Pavard, J.-C., Rolland, A., Schweizer, M., and Singer, D.: Foraminifères estuariens de la façade atlantique française : Guide de détermination, Zenodo, https://doi.org/10.5281/zenodo.14197875, 2023.
Keeley, N. B., Macleod, C. K., and Forrest, B. M.: Combining best professional judgement and quantile regression splines to improve characterisation of macrofaunal responses to enrichment, Ecol. Indic., 12, 154–166, https://doi.org/10.1016/j.ecolind.2011.03.022, 2012.
Klootwijk, A. T. and Alve, E.: Does the analysed size fraction of benthic foraminifera influence the ecological quality status and the interpretation of environmental conditions? Indications from two northern Norwegian fjords, Ecol. Indic., 135, 108423, https://doi.org//10.1016/j.ecolind.2021.108423, 2022.
Kütter, V. T., Almeida Moreira, V., Tavares Kütter, M., Vieira Silva-Filho, E., Duarte Marques, E., Garnier, J., and Dausacker Bidone, E.: Metal bioavailability and distribution in the fish community in a tropical estuary, Sepetiba Bay, Rio De Janeiro, Brazil, Geochimica Brasiliensis, 351, 17–38, https://doi.org/10.21715/GB2358-2812.2021351017, 2021.
Labrune, C., Amouroux, J.-M., Sarda, R., Dutrieux, E., Thorin, S., Rosenberg, R., and Grémare, A.: Characterization of the ecological quality of the coastal Gulf of Lions (NW Mediterrenean). A comparative approach based on three biotic indices, Mar. Pollut. Bull., 52, 34–47, 2006.
Landis, J. R. and Koch, G. G.: The measurement of observer agreement for categorical data, Biometrics, 33, 159–174, 1977.
Laut, L., Martins, M. V. A., Frontalini, F., Ballalai, J. M., Belart, P., Habib, R., Fontana, L. F., Clemente, I. M. M. M., Lorini, M. L., Filho, J. G. M., Laut, V. M., and Figueiredo, M. S. L.: Assessment of the trophic state of a hypersaline-carbonatic environment: Vermelha Lagoon (Brazil), PLoS ONE, 12, e0184819, https://doi.org/10.1371/journal.pone.0184819, 2017.
Laut, L., Matta, G., Camara, G., Belart, P., Clemente, I., Ballalai, J., Volino, E., and Couto, E. C. G.: Living and dead foraminifera assemblages as environmental indicators in the Almada River Estuary, Ilhéus, northeastern Brazil, J. S. Am. Earth Sci., 105, 102883, https://doi.org/10.1016/j.jsames.2020.102883, 2021.
Laut, L., Belart, P., Carelli, T., Martins, M. V. A., and Laut, V.: Insights into the Ecology of Foraminifera from the Most Hypersaline Lagoon in Brazil: Vermelha Lagoon, Estuar. Coast., 45, 2632–2649, https://doi.org/10.1007/s12237-022-01073-x, 2022.
Laut, L. L. M., Martins, V., da Silva, F. S., Crapez, M. A. C., Fontana, L. F., Carvalhal-Gomes, S. B. V., and Souza, R. C. C. L.: Foraminifera, thecamoebians, and bacterial activity in polluted intertropical and subtropical Brazilian estuarine systems, J. Coastal Res., 32, 56–69, 2016.
Lavesque, N., Blanchet, H., and de Montaudouin, X.: Development of a multimetric approach to assess perturbation of benthic macrofauna in Zostera noltii beds, J. Exp. Mar. Biol. Ecol., 368, 101–112, 2009.
Leong, L. S. and Tanner, P. A.: Comparison of methods for determination of organic carbon in marine sediment, Mar. Pollut. Bull., 38, 875–879, 1999.
Luczak, C., Janquin, M.-A., and Kupka, A.: Simple standard procedure for the routine determination of organic matter in marine sediment, Hydrobiologia, 345, 87–94, 1997.
Martins, M. V. A., Zaaboub, N., Aleya, L., Frontalini, F., Pereira, E., Miranda, P., Mane, M., Rocha, F., Laut, L., and El Bour, M.: Environmental Quality Assessment of Bizerte Lagoon (Tunisia) Using Living Foraminifera Assemblages and a Multiproxy Approach, Plos One, 10, e0137250, https://doi.org/10.1371/journal.pone.0137250, 2015.
Martins, M. V. A., Hohenegger, J., Martiìnez-Coloìn, M., Frontalini, F., Bergamaschi, S., Laut, L., Belart, P., Mahiques, M., Pereira, E., Rodrigues, R., Terroso, D., Miranda, P., Geraldes, M. C., Villena, H. H., Reisi, T., Socorro, O. A. A., Sousa, S. H. M., Yamashita, C., and Rocha, F.: Ecological quality status of the NE sector of the Guanabara Bay (Brazil): A case of living benthic foraminiferal resilience, Mar. Pollut. Bull., 158, 111449, https://doi.org/10.1016/j.marpolbul.2020.111449, 2020.
McLusky, D. S. and Elliott, M.: Transitional waters: a new approach, semantics or just muddying the waters?, Estuar. Coast. Shelf S., 71, 359–363, 2007.
Melis, R., Celio, M., Bouchet, V. M. P., Varagona, G., Bazzaro, M., Crosera, M., and Pugliese, N.: Seasonal Response of benthic foraminifera to anthropogenic pressure in two stations of the Gulf of Trieste (northern Adriatic Sea): the marine protected area of Miramare versus the Servola water sewage outfall, Mediterr. Mar. Sci., 20, 120–141, 2019.
Muniz, P., Venturini, N., Pires-Vanin, A. M. S., Tommasi, L. R., and Borja, A.: Testing the applicability of a Marine Biotic Index (AMBI) to assessing the ecological quality of soft-bottom benthic communities, in the South America Atlantic region, Mar. Pollut. Bull., 50, 624–637, 2005.
Muniz, P., Venturini, N., Hutton, M., Kandratavicius, N., Pita, A., Brugnoli, E., Burone, L., and García-Rodríguez, F.: Ecosystem health of Montevideo coastal zone: A multi approach using some different benthic indicators to improve a ten-year-ago assessment, J. Sea Res., 65, 38–50, 2011.
Nunes, M., Martins, M. V. A., Frontalini, F., Bouchet, V. M. P., Francescangeli, F., Hohenegger, J., Figueira, R., Senez-Mello, T. M., Castelo, W. F. L., Damasceno, F. L., Laut, L., Duleba, W., Sousa, S. H. M., Antonioli, L., and Geraldes, M. C.: Inferring the ecological quality status based on living benthic foraminiferal indices in transitional areas of the Guanabara bay (SE Brazil), Environ. Pollut., 320, 121003, https://doi.org/10.1016/j.envpol.2023.121003, 2023.
O'Brien, P., Polovodova Asteman, I., and Bouchet, V. M. P.: Benthic foraminiferal indices and environmental quality assessment of transitional waters: a review of current challenges and future research perspectives, Water, 13, 1898, https://doi.org/10.3390/w13141898, 2021.
O'Malley, B. J., Schwing, P. T., Martínez-Colón, M., Spezzaferri, S., Machain-Castillo, M. L., Larson, R. A., Brooks, G. R., Ruiz-Fernandez, A. C., and Hollander, D. J.: Development of a benthic foraminifera based marine biotic index (Foram-AMBI) for the Gulf of Mexico: A decision support tool, Ecol. Indic., 120, 106916, https://doi.org/10.1016/j.ecolind.2020.106916, 2021.
Parent, B., Barras, C., Bicchi, E., Charrieau, L. M., Choquel, C., Bénéteau, É., Maillet, G. M., and Jorissen, F. J.: Comparison of Four Foraminiferal Biotic Indices Assessing the Environmental Quality of Coastal Mediterranean Soft Bottoms, Water, 13, 3193, https://doi.org/10.3390/w13223193, 2021.
Pavard, J.-C., Richirt, J., Seuront, L., Blanchet, H., Fouet, M. P. A., Humbert, S., Gouillieux, B., Duong, G., and Bouchet, V. M. P.: The great shift: the non-indigenous species Ammonia confertitesta (Foraminifera, Rhizaria) outcompete indigenous Ammonia species in the Gironde estuary (France), Estuar. Coast. Shelf S., 289, 108378, https://doi.org/10.1016/j.ecss.2023.108378, 2023.
Pusceddu, A., Dell'Anno, A., Danovaro, R., Manini, E., Sara, G., and Fabiano, M.: Enzymatically hydrolyzable protein and carbohydrate sedimentary pools as indicators of the trophic state of detritus sink systems: a case study in a Mediterranean coastal lagoon, Estuaries, 26, 641–650, 2003.
Raposo, D., Clemente, I., Figueiredo, M., Vilar, A., Lorini, M. L., Frontalini, F., Martins, M. V. A., Belart, P., Fontana, L., Habib, R., and Laut, L.: Benthic foraminiferal and organic matter compounds as proxies of environmental quality in a tropical coastal lagoon: The Itaipu lagoon (Brazil), Mar. Pollut. Bull., 129, 114–125, https://doi.org/10.1016/j.marpolbul.2018.02.018, 2018.
Raposo, D., Frontalini, F., Clemente, I., Couto, E. d. C. G., Veriìssimo, F., and Laut, L.: Benthic Foraminiferal Response to Trace Elements in a Tropical Mesotidal Brazilian Estuary, Estuar. Coast., 45, 2610–2631, 2022.
Ribeiro, A. P., Figueiredo, A. M. G., Santos, J. O. d., Ferreira, P. A. d. L., Graudenz, G. S., Ruiz, M. S., Mahiques, M. M. d., Figueira, R. C. L., and Wasserman, J. C. F. A.: Effects of contamination with toxic metals on the environmental quality of Sepetiba Bay SE Brazil: The case of Ingá Company, Management of Environmental Quality, 26, 538–551, https://doi.org/10.1108/MEQ-06-2013-0074, 2015.
Rodrigues, A. R., Díaz, T. L., and Pellizari, V. H.: Living Foraminifera in a Brazilian Subtropical Coastal Environment (Flamengo Inlet, Ubatuba, São Paulo State–Brazil), in: Approaches to Study Living Foraminifera, edited by: Kitazato, H. and Bernhard, J. M., Environmental Science and Engineering, Springer, Tokyo, 195–227, https://doi.org/10.1007/978-4-431-54388-6_11, 2014.
Rovai, A. S., Twilley, R. R., Worthington, T. A., and Riul, P.: Brazilian mangroves: blue carbon hotspots of national and global relevance to natural climate solutions, Frontiers in Forests and Global Change, 4, 787533, https://doi.org/10.3389/ffgc.2021.787533, 2022.
Rudorff, N. M., Bonetti, C., and Bonetti Filho, J.: Suspended shellfish culture impacts on the benthic layer: a case study in Brazilian subtropical waters, Braz. J. Oceanogr., 60, 221–234, 2012.
Schintu, M., Marrucci, A., Marras, B., Galgani, F., Buosi, C., Ibba, A., and Cherchi, A.: Heavy metal accumulation in surface sediments at the port of Cagliari (Sardinia, western Mediterranean): environmental assessment using sequential extractions and benthic foraminifera, Mar. Pollut. Bull., 11, 45–56, 2016.
Schönfeld, J., Alve, E., Geslin, E., Jorissen, F., Korsun, S., Spezzaferri, S., Abramovich, S., Almogi-Labin, A., Armynot du Chatelet, E., Barras, C., Bergamin, L., Bicchi, E., Bouchet, V. M. P., Cearreta, A., Di Bella, L., Dijkstra, N., Disaró, S. T., Ferraro, L., Frontalini, F., Gennari, G., Golikova, E., Haynert, K., Hess, S., Husum, K., Martins, V., McGann, M., Oron, S., Romano, E., Mello Sousa, S., and Tsujimoto, A.: The FOBIMO (FOraminiferal BIo-MOnitoring) initiative – Towards a standardised protocol for soft-bottom benthic foraminiferal monitoring studies, Mar. Micropaleontol., 94–95, 1–13, 2012.
Simboura, N. and Zenetos, A.: Benthic indicators to use in ecological quality classification of Mediterranean soft bottom marine ecosystems, including a new biotic index, Mediterr. Mar. Sci., 3, 77–111, 2002.
Simpson, G. L. and Oksanen, J.: analogue: Analogue and weighted averaging methods for palaeoecology, R-Project.org [code], https://doi.org/10.32614/CRAN.package.analogue, 2021.
Sokal, R. R. and Rohlf, F. J.: Biometry, W. H. Freeman, ISBN-13 978-0-7167-2411-7, 1995.
Sousa, S., Yamashita, C., Semensatto, D. J., Santarosa, A., Iwai, I., Omachi, C., Disaró, S. T., Martins, M. V., Barbosa, C., Bonetti, C., Vilela, C., Laut, L., Turra, T., Alve, E., Armynot du Châtelet, E., V.M.P., B., Burone, L., Carmo, A., Figueira, R., Figueiredo, M., Frontalini, F., Geslin, E., Lamparelli, C., Licari, L., Lorini, M., Lourenço, R., Mahiques, M., Martin, T., Mello, R., Millo, C., Moreira, D., Pawlowski, J., Rodrigues, A., and Siegle, E.: Opportunities and challenges in incorporating benthic foraminifera in marine and coastal environmental biomonitoring of soft sediments: from science to regulation and practice, Journal of Sedimentary Environments, 5, 257–265, 2020.
Souza, A. M., Rocha, D. S., Guerra, J. V., Cunha, B. A., Martins, M. V. A., and Geraldes, M. C.: Metal concentrations in marine sediments of the Rio de Janeiro Coast Brazil: A proposal to establish new acceptable levels of contamination, Mar. Pollut. Bull., 165, 112113, doi.org/10.1016/j.marpolbul.2021.112113, 2021a.
Souza, F. M., Gilbert, E. R., Brauko, K. M., Lorenzi, L., Machado, E., and Camargo, M. G.: Macrobenthic community responses to multiple environmental stressors in a subtropical estuary, PeerJ, 9, e12427, https://doi.org/10.7717/peerj.12427, 2021b.
Stal, L. J.: Cyanobacterial mats and stromatolites, in: Ecology of Cyanobacteria II: Their Diversity in Space and Time, edited by: Whitton, A. B., Springer, Dordrecht, 65–125, https://doi.org/10.1007/978-94-007-3855-3_4, 2012.
Ter Braak, C. J. F.: Unimodal Models to Relate Species to Environment, University of Wageningen, https://edepot.wur.nl/201452 (last access: 14 August 2025), 1987.
Valença, A. P. and Santos, P. J.: Macrobenthic community for assessment of estuarine health in tropical areas (Northeast, Brazil): review of macrofauna classification in ecological groups and application of AZTI Marine Biotic Index, Mar. Pollut. Bull., 64, 1809–1820, 2012.
Veileder: Klassifisering av miljøtilstand i vann, https://www.vannportalen.no/veiledere/klassifiseringsveileder/ (last access: 14 August 2025), 2018.
Viaroli, P., Bartoli, M., Giordani, G., Magni, P., and Welsh, D. T.: Biogeochemical indicators as tools for assessing sediment quality/vulnerability in transitional aquatic ecosystems, Aquat. Conserv., 14, 19–29, https://doi.org/10.1002/aqc.647, 2004.
Vilela, C. G., Batista, D. S., Baptista-Neto, J. A., Crapez, M., and McAllister, J. J.: Benthic foraminifera distribution in high polluted sediments from Niterói Harbor (Guanabara Bay), Rio de Janeiro, Brazil, An. Acad. Bras. Cienc., 76, 161–171, 2004.
Zettler, M. L., Proffitt, C. E., Darr, A., Degraer, S., Devriese, L., Greathead, C., Kotta, J., Magni, P., Martin, G., Reiss, H., Speybroeck, J., Tagliapietra, D., Van Hoey, G., and Ysebaert, T.: On the myths of indicator species: issues and further consideration in the use of static concepts for ecological applications, Plos One, 8, e78219, https://doi.org/10.1371/journal.pone.0078219, 2013.