the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
The influence of three common antibiotics on coastal benthic foraminifera: implications for culture experiments and biomonitoring
Michael Lintner
Irina Polovodova Asteman
Wolfgang Wanek
Petra Heinz
Jan Goleń
Jarosław Tyszka
Synthetic antibiotics are medicinal substances crucial for human and animal health and welfare. Recently they have been expansively used in the food industry for reducing bacterial infections in livestock, poultry, and aquaculture. Due to their extensive use, antibiotics are increasingly accumulating in coastal marine ecosystems and cause damage to marine organisms. In this study we investigated the influence of antibiotics on benthic foraminifera, which are widespread marine protists. Foraminifera are often used as bioindicators to define the health state of coastal ecosystems. To gain deeper insights into the ecology of foraminifera and enhance their use as bioindicators, numerous studies have conducted laboratory experiments, with some employing antibiotics to prevent bacterial infections in the cultures. However, for decades it remained unresolved whether antibiotics have either a negative or a positive effect on foraminifera. In this study we tested the influence of three commonly used antibiotics (ampicillin, chloramphenicol, and tetracycline) as well as a mixture of the three on nutrient uptake of two benthic foraminifera, temperate fjord species Nonionella sp. T1 and large tropical species Heterostegina depressa. Our results showed that tetracycline present alone or in mixture has the most negative influence on the nutrition uptake of foraminifera, and under light conditions it may completely inactivate foraminiferal activity. Ampicillin showed a less negative impact, likely caused by a hydrolysis of this drug in seawater over days. Finally, chloramphenicol reduced the nutrient uptake of the symbiont-bearing H. depressa but not that of Nonionella sp. T1, which indicates that this antibiotic exerts a species-specific effect. However, given that the applied antibiotic concentrations were high following the supplier's recommendation for laboratory cultures, an extrapolation of these results to antibiotic concentrations occurring in coastal waters is difficult.
- Article
(2061 KB) - Full-text XML
- BibTeX
- EndNote
1.1 Antibiotics as pollutants in aquatic microenvironments
Antibiotics are bioactive chemical compounds which act against bacteria either by killing them (bactericidal antibiotics) or by inhibiting their growth (bacteriostatic antibiotics). Nowadays, antibiotics are extensively used in both human and veterinary medicine, causing both their release with sewer outlets and their subsequent accumulation in the environment. Other areas of antibiotics application such as agriculture, livestock, and aquaculture cause the direct introduction of antibiotics into terrestrial and aquatic ecosystems. Antibiotics can enter the hydrosphere both as unaltered parent compounds and as antibiotic metabolites (Gothwal and Shashidhar, 2015). In fact, between 50 %–80 % of the ingested antibiotics are excreted by organisms unaltered and enter the environment through faeces and urine (Lienert et al., 2007). As a result, antibiotics reach the sea indirectly via rivers and groundwater from agricultural and urban point sources (García-Galán et al., 2010) or are directly introduced in aquaculture (Adenaya et al., 2023). Concentrations of antibiotics in groundwater can range from a few ng L−1 to several µg L−1 (Gros et al., 2021). For instance, China is the world's largest producer and consumer of antibiotics (Zhang et al., 2015), with around 2.5×104 t of antibiotics being consumed annually (Bu et al., 2013), which causes a strong accumulation of antibiotics in the Chinese coastal zones (Wang et al., 2023). The negative influence of antibiotics in seawater has been studied for several marine organisms. For instance, the water flea Daphnia magna sensitively reacts to the presence of antibiotics and shows increased mortality at increased concentrations (Bawa-Allah and Ehimiyein, 2022). Further, antibiotics have negative side effects like ototoxicity, nephrotoxicity, or tendinopathy in mammalian cells (Khaliq and Zhanel, 2003 ) and can lead to dysfunction and oxidative damage in eukaryotic cells (Kalghatgi et al., 2013). Further, antibiotics can accumulate in organisms (bioaccumulation) and transfer via the food chain (Hu et al., 2023). In summary, the presence of antibiotics in surface water, groundwater, and seawater causes the exposure of (micro)biota to these pollutants, either directly or through trophic interactions and feeding activities, as antibiotics can also accumulate in plants and thus find their way into the food chain of higher organisms (Gothwal and Shashidhar, 2015).
1.2 Foraminifera and antibiotics
Foraminifera are marine protists found in all marine habitats in high abundance. In the late 1960s it was suggested that the presence of bacteria in the culture medium affects the growth of foraminifera under laboratory conditions (Müller and Lee, 1969). First attempts at removing bacteria as ectobionts from foraminifera by antibiotic treatment resulted in the failed growth of foraminifera (e.g. Lee and Pierce, 1963), whilst later studies demonstrated that streptomycin increases the growth of foraminifera (Nigam et al., 1996). Yet, Nigam et al. (1996) only tested the impact of one antibiotic type on the growth of foraminifera but did not investigate the potential effects on their nutritional and metabolic activity. Interestingly, the study of Nigam et al. (1996) remains relevant as – due to the initial widespread use of streptomycin in medicine – many bacterial species have evolved or acquired streptomycin resistance (e.g. Cohen et al., 2020). Some studies also tried to address how mixtures of different antibiotics affect foraminifera under laboratory conditions. For instance, a mixture of streptomycin, penicillin, and amphotericin B was shown to inhibit the growth of multiple foraminifera (Lee et al., 1991), whilst chloramphenicol and streptomycin (Arnold, 1966) or chloramphenicol, dihydrostreptomycin, neomycin, penicillin, and tetracycline (Pierce, 1965) did not inhibit growth and reproduction across several foraminiferal species. The latter approach was adopted by experiments addressing denitrification in benthic foraminifera and requiring bacteria-free cultures (e.g. Bernhard et al., 2012). Nowadays many studies on foraminifera are carried out using antibiotics, though the impact of antibiotics on the nutritional and metabolic activity of the foraminifera has barely been studied.
At present, a variety of different antibiotics, which have different mechanisms of action and therefore also affect different bacteria, are in use. In this study we investigated the effects of three antibiotics commonly used in human and animal treatment, which are also frequently used in cell cultures. Those include (1) ampicillin (AMP), (2) chloramphenicol (CAM), and (3) tetracycline (TET).
-
Ampicillin is classified as a beta-lactam, which consists of a highly reactive ring made up of three carbon atoms and one nitrogen atom. Through the interaction of this ring with proteins of the bacterial cell wall, the synthesis of the cell wall is disrupted and the growth of the bacteria is inhibited (Etebu and Arikekpar, 2016). Under light protection, AMP degrades slowly following a temperature-dependent pattern, e.g. within 24 h, 6% of AMP degrades at 4 °C and 10 % at 23 °C (Zhang and Trissel, 2002). AMP degradation is also pH sensitive, with half-life times decreasing from 31 d to 27 and 7 d when pH increased from 4 to 7 and 9, respectively (Mitchell et al., 2014).
-
Chloramphenicol is a 50S ribosome inhibitor and physically blocks the initiation phase of protein translation in bacteria (Patel and Bonomo, 2011). CAM has a low water solubility (2.5–4.4 mg mL−1 at 25 °C) and decomposes rapidly through hydrolysis at pH < 9.5 (Shaw, 1975). The CAM molecule is relatively temperature insensitive, with only 50 % of CAM being inactivated at 37 °C during 6 months (Shaw, 1975).
-
Tetracycline disrupts the protein synthesis in bacteria by preventing the elongation of polypeptide chains through curtailing the amino acid addition (Etebu and Arikekpar, 2016). With 10 mg mL−1 the water solubility is intermediate and comparable with CAM (Ali, 1984). TET quickly degrades in acidic solutions in the pH range 2 to 7, whilst above pH = 7, TET is relatively stable (Ali, 1984). However, the biggest challenge with this antibiotic is its sensitivity to visible light. After 2 h of light exposure, 27 % of the initial amount of tetracycline is degraded (Oluwole and Olatunji, 2022).
In our study we investigate the influence of the antibiotics AMP, CAM, and TET as well as their mixture (MIX) on the nutrition of foraminifera. We studied two benthic foraminiferal species from two different habitats. Heterostegina depressa d'Orbigny, a large shallow-water tropical benthic foraminifer with algal endosymbionts, was incubated in light conditions and at 25 °C. Nonionella sp. T1, on the other hand, is a temperate species, which was incubated in dark conditions and at 10 °C because it comes from a ∼40 m deep fjord setting. Through this combination, the sensitivity of antibiotics to both light and temperature was investigated in this study.
2.1 Sediment sampling and sampling area
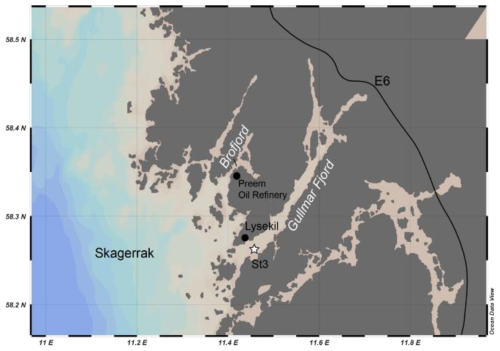
Sediment sampling in the Gullmar Fjord for living Nonionella sp. T1 was performed on 2 October 2023 aboard R/V Alice (University of Gothenburg) by using a box corer at station 3 in the outer part of the Gullmar Fjord (58°15.538′ N, 11°27.483′ E, and 40 m water depth: Fig. 1).
Gullmar Fjord is located on the west coast of Sweden, with a maximum water depth of 118.6 m. It is one of the most well-studied marine settings in the world, with the first hydrographic observations taken as early as 1869 (Ekman, 1870). Being a sill fjord, Gullmar Fjord has remarkably high sediment accumulation rates of 0.7–1.4 cm yr−1 (Filipsson and Nordberg, 2004) and low tidal activity providing a high-resolution paleo-environmental archive (e.g. Harland et al., 2013; Polovodova Asteman et al., 2018). Due to its high biodiversity, Gullmar Fjord became the first marine conservation area in Sweden in 1983 and currently has no anthropogenic impact.
In parallel to the sediment sampling, environmental data (temperature, salinity, and oxygen) were taken aboard by using a CTD probe. The T, S, and O2 observations proximal to the sea floor showed a temperature of 14.9 °C, salinity of 33, and 4.1 mL O2 L−1, respectively. The retrieved sediment surface in the box corer was intact and covered by water, which was gently siphoned out by using a plastic tube. The upper 5 cm of the sediment was transferred to a bucket and taken to the lab at the Kristineberg Centre for Marine Research and Innovation. There the sediment was gently sieved using 63 and 1000 µm sieves and ambient fjord water to concentrate living foraminifera and remove larger meiofauna. The resulting sediment fraction (63–1000 µm) with living Nonionella sp. T1 was collected in wide-neck PVC bottles and sent to the Institute of Geological Sciences, Polish Academy of Sciences (ING PAN), Kraków, for experimental work.
Sediments containing the large benthic foraminifera Heterostegina depressa come from a main culture, hosted at the Department of Palaeontology of the University of Vienna for many years, which originally comes from a shark tank in the Haus der Natur located in the city of Salzburg (Austria). In Vienna, H. depressa is continuously cultivated at a temperature of 25 °C, a salinity of 35 PSU, and a light intensity of 40 µmol photons m−2 s−1 in an 8–16 h light–dark rhythm. The sediment with living H. depressa was sent to the ING PAN Research Center in Kraków to run the experiments in the Biogeosystem Modelling Lab (BioGeoLab).
2.2 Experimental setup
All cultivation experiments were carried out at the ING PAN BioGeoLab in Kraków. After the sediment arrived from the different sampling locations, two separate main cultures were created. For that, the sediment was placed in glass aquaria and covered with artificial seawater (salinity of 33 for Gullmar Fjord and 35 for Vienna sediments). In alignment with the lifestyles of the two selected species, the aquarium containing the Gullmar Fjord sediment was placed in the dark at 15 °C, whilst the Vienna sediment was placed at room temperature (25 °C) and was illuminated with an 8 h : 16 h light cycle. After a week-long acclimatization phase, the sediment was washed over a 150 µm sieve, and living foraminifera, which had a characteristic brown-yellow colour of the cytoplasm, were removed using a fine brush. Nonionella sp. T1 was collected from the Gullmar Fjord sediments and H. depressa from the Vienna culture. Following that, all foraminifera were cleaned with a brush, and all adhering particles were carefully removed and transferred to a crystallization dish, placed in the centre of the dish, and left for 1 d to test their vitality by a so-called “crawling test”. For this purpose, the foraminifera are placed centrally in a petri dish. After a few hours, those foraminifera that have moved away from the centre and thus showed pseudopodia activity were used for the experiments. After 1 d, only the actively moving individuals were selected for further feeding experiments.
For feeding and isotopic uptake experiments, 15 individuals of Nonionella sp. T1 and 1 individual of H. depressa were placed for each crystallization dish and covered with 100 mL artificial seawater with a salinity similar to that observed at the sampling sites. The cultures without antibiotics served as control samples. The experiments with Nonionella sp. T1 were performed in triplicates, whilst for H. depressa six replicates were used. Different replication was done in order to get a final mass of 1 mg dry weight of cytoplasm for further analysis. As interference factor three antibiotics (ampicillin, chloramphenicol, and tetracycline) were used in two concentrations and additionally as a mixture containing all three antibiotics together. The low concentration (c1) was based on the recommendation from the providing company (Roth) to keep the culture medium free of bacterial activity. Therefore, we created four culture media on the c1 level separately enriched with 30 µg mL−1 AMP (ampicillin), 50 µg mL−1 CAM (chloramphenicol), and 100 µg mL−1 TET (tetracycline), and a mixture (MIX) of 30 µg mL−1 AMP, 50 µg mL−1 CAM, and 100 µg mL−1 TET altogether. To see an effect of the increasing concentration we further created four culture media (c2) with 10-times-higher levels of antibiotics: 300 µg mL−1 AMP, 500 µg mL−1 CAM, 1000 µg mL−1 TET, and a MIX of the same respective concentrations.
To observe the change in the metabolic activity of the foraminifera, lyophilized isotopic labelled alga powder (Chaetoceros simplex var. calcitrans) was added to the cultures with Nonionella sp. T1. This diatom species also occurs naturally in the Gullmar Fjord (Hällfors, 2004). Chaetoceros simplex var. calcitrans was grown in a nutrient medium enriched with 13C and 15N and therefore contains an increased concentration of isotopes (for method details see Lintner et al., 2020). Since H. depressa with its symbionts is fully autotrophic (Röttger et al., 1980), we directly spiked the culture medium containing these foraminifera with NaH13CO3 (0.2 mM) and Na15NO3 (0.2 mM) to investigate the isotopic uptake. Following this, the experimental setups for both species were incubated with antibiotics for 3 and 7 d.
2.3 Sample and data processing
After incubation with the isotopic enriched algae, Nonionella sp. T1 individuals were removed from the culture and cleaned of any adhering particles using a brush. To measure the isotopic uptake, 15 specimens of Nonionella sp. T1 and 1 specimen of H. depressa were transferred to separate Sn capsules. In total, 48 capsules for Nonionella sp. T1 (4 antibiotics × 2 concentrations × 2 time points × 3 replicates) and 96 capsules for H. depressa (4 antibiotics × 2 concentrations × 2 time points × 6 replicates) were used. Afterwards, the capsules including foraminifera were dried for 3 d at room temperature. The calcitic test was then removed by adding 12 µL of 4 % hydrochloric acid (HCl) to each capsule. Finally, the capsules were dried at 50 °C for 3 d and sent to the University of Vienna for further measurements. The isotope mass ratio was measured at the Stable Isotope Laboratory for Environmental Research (SILVER – University of Vienna; IRMS, DeltaPLUS, coupled by a ConFlo III interface to an elemental analyser EA 1110, Thermo Finnigan, was used to measure ratios of 13C 12C and 15N 14N by using Vienna PeeDee Belemnite Standard RVPDB=0.0112372 for C and atmospheric nitrogen RatmN=0.0036765 for N), and the calculation of the amount of phytodetrital isotopes (pC and pN) for Nonionella sp. T1 versus incorporated isotopes (IC and IN) for H. depressa was done according to Lintner et al. (2020). To investigate the influence of antibiotics on the metabolism of foraminifera, the pC : pN (IC : IN) ratio is calculated to determine whether the foraminifera are in a stressed state or not (Lintner et al., 2025). Two-way analysis of variance (ANOVA) tests (level of significance p=0.05) using PAST4.0 software (Hammer and Harper, 2001) were performed to test whether the type of antibiotics, their concentration, and their incubation time significantly affected the food uptake in both species. Further, a post hoc test (Tukey's post hoc test) was applied to test the differences between the used antibiotics and the control sample.
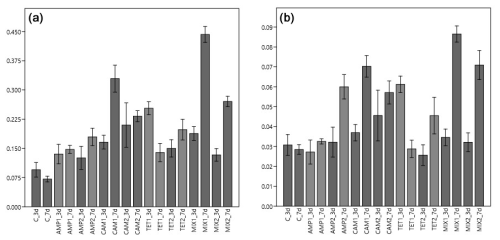
Figure 2Carbon (pC, a) and nitrogen (pN, b) uptake from Nonionella sp. T1 after three (3 d) and seven (7 d) days of incubation. The abbreviations indicate the control (C) sample or the antibiotics added during incubation: AMP – ampicillin; CAM – chloramphenicol; TET – tetracycline; MIX – mixture of all three used antibiotics (AMP, CAM, and TET). 1 is the manufacturer's recommended concentration to remove bacterial activity in the culture medium, and 2 corresponds to 10 times the concentration of 1.
Table 1Post hoc test of carbon (grey) and nitrogen (white) uptake in Nonionella sp. T1 and H. depressa. Significant values are shown in bold. Control indicates foraminifera incubated without any antibiotics. The abbreviations indicate the antibiotics present during incubation: AMP – ampicillin; CAM – chloramphenicol; TET – tetracycline; MIX – mixture of all three used antibiotics (AMP, CAM, and TET).
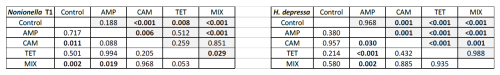
Our first experimental setup with Nonionella sp. T1 showed that food (carbon and nitrogen) uptake of this species was highly affected by the presence of antibiotics (p<0.001), their concentration (p<0.001), and their interaction with each other (p<0.001). The post hoc test of the incorporated amount of carbon (pC) highlights the difference between the digested food in each experiment. The control group metabolized significantly less carbon compared to the foraminifera incubated with CAM, TET, or MIX (p values provided in Table 1). However, no difference (p=0.188) in carbon uptake was observed between the control group and foraminifera incubated with AMP. In all experiments with Nonionella sp. T1 the mean values of the incorporated carbon (pC) were higher if antibiotics were present in the culture medium (Fig. 2). The carbon uptake in the AMP spiked samples was not affected by the concentration (p=0.640) of the antibiotics or by time (p=0.190). Also, the concentration of CAM did not appear to influence the pC (p=0.476), but here the amount of pC increased significantly (p=0.003) with time. Similar to AMP, the TET concentration (p=0.351) and the incubation time (p=0.183) did not affect the carbon uptake in this treatment. If all antibiotics were present in the culture medium (MIX), the concentration of the antibiotics significantly (p<0.001) decreased pC, and also significantly (p<0.001) more carbon was incorporated with time.
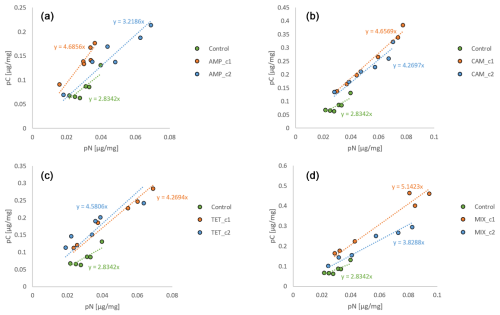
Figure 3pC : pN ratios from Nonionella sp. T1. The abbreviations indicate the antibiotics added during incubation: AMP – ampicillin (a); CAM – chloramphenicol (b); TET – tetracycline (c); MIX – mixture (d) of all three used antibiotics (AMP, CAM, and TET). c1 is the manufacturer's recommended concentration to remove bacterial activity in the culture medium, and c2 corresponds to 10 times the concentration of c1.
The nitrogen uptake (pN) for Nonionella sp. T1 showed a different pattern. No significant difference in pN was observed between the control group and AMP (p=0.717) or TET (p=0.501) group, but the presence of CAM (p=0.011) or MIX (p=0.002) significantly increased pN in this species (Table 1). Between 3 and 7 d no significant difference (p=0.701) was observed in pN of the control group. The concentration of AMP in the culture medium appeared to play a significant role (p=0.023), as did the time of incubation (p=0.021). At a low level (AMP_c1) this antibiotic did not influence pN during the incubation (p=0.437), but at c2 a significant increase (p=0.046) of pN was noticed during the incubation (Fig. 2). For the presence of CAM in the culture medium with Nonionella sp. T1 an opposite trend was observed. Here the lower concentration c1 led to an increase in pN with time (p=0.008), and at c2 no significant difference (p=0.454) was observed between 3 and 7 d of incubation (Fig. 2). Finally, for TET, its low concentration (c1) decreased the pN with time (p=0.006), whilst at TET c2 no significant influence (p=0.134) was observed in pN between day three and seven. In both c1 and c2 concentrations the presence of all three antibiotics together (MIX) led to an increase in pN with time (c1: p<0.001; c2: p=0.11) (Fig. 2).
To investigate the influence of antibiotics on the overall metabolism of foraminifera, the ratio of pC : pN was used (Fig. 3). This ratio must always be seen in comparison to the control sample. Generally, the higher the slope value, the better the foraminiferal fitness during the experiment is, since a low value means an increase in pN, which is equivalent to the production of stress proteins (Lintner et al., 2025). All control samples have the lowest slope (y=2.8342x) in all treatments (Fig. 3), which indicates that the presence of antibiotics contributes significantly to changes in the metabolism of the foraminifera. In the AMP, CAM, and MIX experiments the slope of c2 is lower than c1, suggesting higher stress for Nonionella sp. T1 caused by a higher level of antibiotics (Fig. 3a, b, and d). Only for the TET experiments is the slope of c1 lower than c2, which correlates with the observation that at c1 pC and pN decrease, in contrast to c2, where both pC and pN increase (Fig. 3c). This highlights that Nonionella sp. T1 incubated with TET likely performs better at its higher concentrations, whereas all other antibiotics harm this species at elevated concentrations.
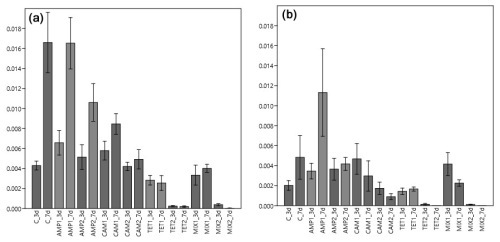
Figure 4Carbon (IC; a) and nitrogen (IN; b) uptake from H. depressa after three (3 d) and seven (7 d) days of incubation. The abbreviation C indicates the control sample. Abbreviations for antibiotics added during incubation: AMP – ampicillin; CAM – chloramphenicol; TET – tetracycline; MIX – mixture of all three used antibiotics (AMP, CAM, and TET). 1 is the manufacturer's recommended concentration to remove bacterial activity in the culture medium, and 2 corresponds to 10 times the concentration of 1.
For our second experimental setup with Heterostegina depressa, the incorporated amount of carbon (IC) in this species significantly depends on the type of antibiotics (p<0.001), exposure time (p<0.001), and also interaction between different antibiotics (p<0.001). Carbon uptake in the control group was similar to H. depressa incubated with AMP (p=0.968) but significantly differed (p<0.001) from all other treatments (CAM, TET, and MIX) (Table 1). The concentration of AMP did not affect (p=0.056) IC, but for both concentrations c1 and c2 a significant increase in IC with time was observed (c1: p=0.006; c2: p=0.036). However, a higher concentration of CAM significantly (p=0.008) reduced the carbon uptake by H. depressa. At the higher CAM concentration (c2) no time exposure effect (p=0.515) was observed, and just a trend (p=0.086) for increasing carbon uptake exists for c1 (Fig. 4). Tetracycline drastically reduced the activity of the foraminifera, and its concentration appeared to play a significant (p<0.001) role. At the same time, between 3 and 7 d of incubation for both TET concentrations (c1: p=0.769; c2: p=0.662) no significant difference was observed. At c2 the uptake was close to zero, indicating a total lack of activity (likely suggesting mortality) of H. depressa individuals even at the shortest time of incubation with TET. The same pattern was found for H. depressa incubated with a MIX of all antibiotics. The higher concentration significantly (p<0.001) reduced foraminiferal activity, and the low concentration (c1) showed no significant (p=0.543) effect of time exposure. However, a significant (p=0.010) difference shown as reduction with time was calculated for c2, which could be explained by the fact that after 3 d there were still very low values of IC in H. depressa and after 7 d an absolute zero IC value was measured (Fig. 4). At this point it must also be mentioned that the culture medium, which included TET, had changed colour from slight yellow to dark red over time. This phenomenon was not observed with TET treatment for Nonionella sp. T1 because of their incubation in dark conditions. However, the cultivation of H. depressa occurred under light exposure, causing TET to decompose, which likely created toxic decomposition products.
The amount of nitrogen (IN) incorporated by H. depressa was also highly affected (p<0.001) by the presence of antibiotics, whilst it was not affected by the incubation time (p=0.293) but rather by their interaction (p=0.045) (Fig. 4). While the concentration of AMP appeared to be of less importance (p=0.1486) in nitrogen processing of H. depressa, all other setups with a higher level of antibiotics reduced the activity of foraminifera significantly (CAM: p=0.039; TET: p<0.001; MIX: p<0.001) (Table 1). The IC : IN ratio was also calculated for H. depressa (Fig. 5). However, the values are subject to a significantly higher spread, which makes them hard to interpret. For Nonionella sp. T1, the values after 3 and 7 d for the pC : pN are on the same trend line, only shifted towards higher values. In H. depressa, the 3 and 7 d values do not plot together, which leads to a decrease in the regression and increases the scatter of data. Antibiotics with a presumably weaker influence, such as AMP or CAM, do not allow for any clear statements about fitness of H. depressa (Fig. 5a and b). However, antibiotics with a clearly negative effect such as TET or MIX also show a shift in the trend line towards a smaller slope, indicating stressful conditions for this species (Fig. 5c and d).
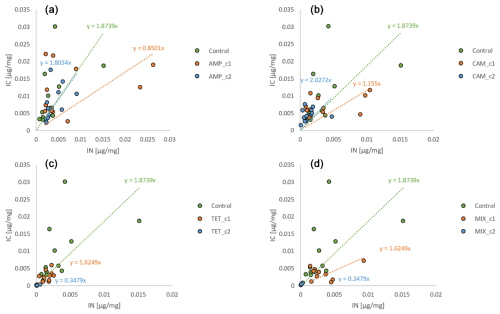
Figure 5IC : IN ratios for H. depressa. The abbreviations indicate the antibiotics added during incubation: IC – incorporated carbon; IN – incorporated nitrogen; AMP – ampicillin (a); CAM – chloramphenicol (b); TET – tetracycline (c); MIX – mixture (d) of all three used antibiotics (AMP, CAM, and TET). c1 is the manufacturer's recommended concentration to remove bacterial activity in the culture medium, and c2 corresponds to 10 times the concentration of c1.
4.1 The response of foraminifera to antibiotics
Based on our results the antibiotic-spiked culture medium appears to cause significant changes in the metabolism of both small and large benthic foraminifera, represented by changes in C : N ratios of the species Nonionella sp. T1 and Heterostegina depressa, respectively. How affected the foraminifera are depends on both the type and concentration of the used antibiotics. The addition of AMP appears to have the weakest impact on both studied species, but at this point it is also questionable whether AMP ever had any influence on the foraminifera. The effective time (time of action before it will decompose) of AMP is around 8 h (Zhang and Trissel, 2002), and it is unstable at high pH values (Mitchell et al., 2014) typical of seawater. Hence, we can assume that the influence of AMP on foraminifera was likely there only within the first few hours of incubation. This is consistent with the low AMP treatment (c1) showing the metabolism of the foraminifera, similar to that of the control group. The high concentration did not have a significant effect on the metabolism of Nonionella sp. T1, but a slight decrease was noticeable in H. depressa. Since c2 was 10 times higher in concentration than c1, it can be assumed that it took longer until there was hardly any active AMP in the culture water. At the same time H. depressa was incubated at higher temperatures, which would cause faster decay of AMP. It is reasonable to believe that Nonionella sp. T1 would be more affected by the higher AMP concentration since its incubation was at lower temperature, but this was not the case. Hence, our results suggest that the effect of ampicillin on the metabolism of foraminifera is highly species dependent.
Our study further shows that the presence of CAM in the culture water did not have a negative effect on Nonionella sp. T1, whereas CAM greatly reduced the metabolism of H. depressa. The hydrolysis of CAM is only significant at high pH values (<9), and this antibiotic is also stable at the temperature range tested here (Shaw, 1975). Based on these assumptions, it can be assumed that both foraminifera interacted with CAM throughout the entire incubation period. Similar to the experiment with AMP, our results once again confirmed that H. depressa is more sensitive to the presence of antibiotics and that the reaction to CAM is species specific as well.
Our results also highlight the strongest difference in metabolism observed for the two studied foraminiferal species in experimental setups with TET and MIX. However, this is not necessarily related to the response of the foraminifera to the antibiotics but can be explained by differences in experimental setups. Since TET (note: also present in MIX treatment) is strongly affected by photodegradation, it is likely that light exposure in H. depressa experiments caused the formation of secondary products, like reactive oxygen species (ROS), which likely negatively affected the foraminifera (Oluwole and Olatunji, 2022). On the other hand, there is a much lower probability of the formation of these secondary products in the culture of Nonionella sp. T1 because of incubations in the dark. Whilst there was also no negative effect on the metabolism of Nonionella sp. T1, we observed a complete deactivation or mortality in H. depressa. This suggests that the secondary products resulting from TET photodegradation can be more toxic to foraminifera than the antibiotic itself.
Finally, we would like to compare our results with previous studies. Based on past data, most studies used streptomycin to investigate its effects on foraminifera. In three (Nigam et al., 1996; Arnold, 1966; Pierce, 1965) of four studies using streptomycin (Pierce, 1965, used dihydrostreptomycin), an increase in foraminifera growth was observed when the antibiotic was present in the culture medium. A positive effect on foraminiferal growth was also observed as soon as CAM was present in the culture medium (Arnold, 1966; Pierce, 1965). We can only partially confirm this result with our data, since CAM has a potential positive effect on the metabolism of Nonionella sp. T1 but a clearly negative influence on H. depressa. Furthermore, Pierce (1965) noticed a positive effect of tetracycline (0.04 %) on growth of foraminifera, but nothing was mentioned about light conditions during the experiment. In our experiments we used 100 and 1000 µg mL−1 TET, which is equal to 0.01 % and 0.1 % and is therefore comparable with the concentrations used by Pierce (1965). However, foraminifera react similarly to TET and to CAM. Again Nonionella sp. T1 was not negatively affected by the antibiotics, but the metabolisms of H. depressa failed completely, which was probably caused by the photodegradation of TET as discussed earlier. In conclusion, it should be mentioned once again that the effect of antibiotics on foraminifera is very species dependent and that the foraminifera used in the laboratory must first be tested for their response to antibiotics before they can be cultivated with them.
4.2 Environmental risk of antibiotics
Apart from the new knowledge provided by this study for foraminifera culture experiments, how can these results be put into an ecological context as mentioned above? Ampicillin is not stable in media with high pH, which is typical of seawater. Accordingly, it is likely that AMP does not pose a direct threat to marine organisms, except in regions where there is a continuously high discharge of this antibiotic into coastal waters. The situation is similar with TET, which, instead of being influenced by the pH, degrades when exposed to light. However, due to the enormous present consumption of TET (e.g. >2500T per year in the European Union) and the low rate of its metabolism in the organisms (up to 75 % will be excreted), tetracycline potentially poses a serious problem in the environment (Daghrir and Drogui, 2013; Xu et al., 2021). It was shown that freshwater algae exhibit toxic effects at TET concentrations > 5 mg L−1, and at 30 mg L−1 they show a 94 % growth inhibition (Xu et al., 2019; Amangelsin et al., 2023). In our experiments, with the TET c1 treatment, despite having significantly higher concentrations (100 mg L−1), we did not observe any negative effect on Nonionella sp. T1, which suggests that some foraminiferal species may be more tolerant to TET than other organisms such as algae. Of all the antibiotics tested, CAM probably has the highest potential to have a negative effect on organisms in the marine environment. In seawater samples from the Herglas Sea in Tunisia, concentrations of 15.6 µg L−1 of CAM were recorded (Tahrani et al., 2016). But apart from these hotspots, common CAM concentrations in seawater are between 0.02 and 0.15 µg L−1 (Nguyen et al., 2022). Those concentrations observed in field by previous studies are now a factor of 1000 lower than those we used in our experiments. However, since the metabolism of our tested foraminifera was only slightly reduced in the presence of CAM (Figs. 2 and 4) and this concentration was artificially high, we cannot assume that in nature there is a negative CAM influence on foraminifera from the currently measured antibiotic concentrations. Additionally, our experiments only provide information about short-term effects on foraminifera caused by antibiotics. To further investigate the resilience of these microeukaryotic organisms, future studies should address whether long-term antibiotic exposure to lower concentrations is more harmful to foraminifera than the exposures tested in this study.
We tested the influence of three antibiotics, ampicillin, chloramphenicol, and tetracycline, and their mixture on the metabolism of two foraminiferal species. Our results show that both species are affected by the presence of antibiotics, but the effects of antibiotics are species specific. In general, Heterostegina depressa responds more sensitively to the presence of antibiotics in culture water than Nonionella sp. T1 – the latter seems to even benefit from the presence of antibiotics. Treatment with ampicillin showed the least negative influence, probably because of a quick ampicillin hydrolysis due to its instability at high pH typical of seawater, and therefore it is likely that ampicillin only interacted with the foraminifera for a few hours. On the other hand, chloramphenicol showed a significant reduction in the metabolism of the foraminifera (especially in H. depressa) indicated by isotopic uptake of carbon and nitrogen. However, the strongest influence in our study was observed for treatments with tetracycline and a mixture of all three antibiotics. Tetracycline reduced the activity of H. depressa to zero within just 3 d (possibly faster), likely caused by its toxic decay products, which arise during incubation with light. In contrast, a negative effect of tetracycline on Nonionella sp. T1 when incubated in dark conditions is hardly noticeable. It is important to note that we have chosen artificially high concentrations of antibiotics in our study to improve future laboratory experiments. To the best of our knowledge, comparably high concentrations in nature are currently not available, which makes it reasonable to believe that the current levels of antibiotics in marine habitats likely do not have a negative impact on foraminifera. However, there is a range of different antibiotics and a high diversity of species, of which we have only tested a few in this study. Hence, future studies must be carried out including several antibiotic types and testing other species to find out which antibiotics have a negative effect on foraminifera and which antibiotics are the best to apply to constrain culture experiments.
Data are available via Zenodo at https://doi.org/10.5281/zenodo.16925684 (Michael, 2025).
ML designed and conducted the experiments, analysed the data, and wrote the manuscript. IPA collected sediment in the fjord and, together with ML, wrote the first draft of the manuscript. WW organized the isotope measurements. PH provided foraminifera from her laboratory. JG assisted with incubation at the ING PAN. JT helped with the study design. All authors read the manuscript carefully.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. While Copernicus Publications makes every effort to include appropriate place names, the final responsibility lies with the authors.
This research is part of project no. UMO-2022/47/P/ST10/01013 co-funded by the National Science Centre and the European Union's Horizon 2020 research and innovation programme under Marie Skłodowska-Curie grant agreement no. 945339. For the purpose of Open Access, the first author has applied a CC-BY public copyright licence to any Author Accepted Manuscript (AAM) version arising from this submission.
This research has been supported by the Narodowym Centrum Nauki (grant no. 945339).
This paper was edited by Malcolm Hart and reviewed by Romana Melis and Takashi Hosono.
Adenaya, A., Berger, M., Brinkhoff, T., Ribas-Ribas, M., and Wurl, O.: Usage of antibiotics in aquaculture and the impact on coastal waters, Mar. Pollut. Bull., 188, 114645, https://doi.org/10.1016/j.marpolbul.2023.114645, 2023.
Ali, S. L.: Tetracycline hydrochloride, in: Analytical profiles of drug substances, Vol. 13, Academic Press, 597–653, https://doi.org/10.1016/S0099-5428(08)60204-X, 1984.
Amangelsin, Y., Semenova, Y., Dadar, M., Aljofan, M., and Bjørklund, G.: The impact of tetracycline pollution on the aquatic environment and removal strategies, Antibiotics, 12, 440, https://doi.org/10.3390/antibiotics12030440, 2023.
Arnold, Z. M.: A laboratory system for maintaining small-volume cultures of foraminifera and other organisms, Micropaleontology, 12, 109–118, 1966.
Bawa-Allah, K. A. and Ehimiyein, A. O.: Ecotoxicological effects of human and veterinary antibiotics on water flea (Daphnia magna), Environ. Toxicol. Pharmacol., 94, 103932, https://doi.org/10.1016/j.etap.2022.103932, 2022.
Bernhard, J. M., Casciotti, K. L., McIlvin, M. R., Beaudoin, D. J., Visscher, P. T., and Edgcomb, V. P.: Potential importance of physiologically diverse benthic foraminifera in sedimentary nitrate storage and respiration, J. Geophys. Res.-Biogeo., 117, G03002, https://doi.org/10.1029/2012JG001949, 2012.
Bu, Q., Wang, B., Huang, J., Deng, S., and Yu, G.:. Pharmaceuticals and personal care products in the aquatic environment in China: a review, J. Hazard. Mater., 262, 189-211, https://doi.org/10.1016/j.jhazmat.2013.08.040, 2013.
Cohen, K. A., Stott, K. E., Munsamy, V., Manson, A. L., Earl, A. M., and Pym, A. S.:. Evidence for expanding the role of streptomycin in the management of drug-resistant Mycobacterium tuberculosis, Antimicrob. Agents Chemother., 64, e00860-20, https://doi.org/10.1128/aac.00860-20, 2020.
Daghrir, R. and Drogui, P.: Tetracycline antibiotics in the environment: a review, Environ. Chem. Lett., 11, 209–227, https://doi.org/10.1007/s10311-013-0404-8, 2013.
Ekman, F. L.: Om salthalten i havsvatten utmed Bohuslänska Kusten. K. Vetenskapsakad, Handlingar, 9, 44 pp., 1870.
Etebu, E. and Arikekpar, I.: Antibiotics: Classification and mechanisms of action with emphasis on molecular perspectives, Int. J. Appl. Microbiol. Biotechnol. Res., 4, 90–101, 2016
Filipsson, H. L. and Nordberg, K.: Climate variations, an overlooked factor influencing the recent marine environment. An example from Gullmar Fjord, Sweden, illustrated by benthic foraminifera and hydrographic data, Estuaries, 27, 867–881, https://doi.org/10.1007/BF02912048, 2004.
García-Galán, M. J., Garrido, T., Fraile, J., Ginebreda, A., Díaz-Cruz, M. S., and Barceló, D.: Simultaneous occurrence of nitrates and sulfonamide antibiotics in two ground water bodies of Catalonia (Spain), J. Hydrol., 383, 93–101, https://doi.org/10.1016/j.jhydrol.2009.06.042, 2010.
Gothwal, R. and Shashidhar, T.: Antibiotic pollution in the environment: a review, Clean-Soil Air Water, 43, 479–489, https://doi.org/10.1002/clen.201300989, 2015.
Gros, M., Catalán, N., Mas-Pla, J., Čelić, M., Petrović, M., and Farré, M. J.: Groundwater antibiotic pollution and its relationship with dissolved organic matter: Identification and environmental implications, Environ. Pollut., 289, 117927, https://doi.org/10.1016/j.envpol.2021.117927, 2021.
Hällfors, G.: Checklist of Baltic Sea Phytoplankton species (including some Heterotrophic Protistan groups), Baltic Sea Environment Proceedings, 9, https://helcom.fi/wp-content/uploads/2019/10/BSEP95.pdf (last access: 28/8/2025), 2004.
Hammer, Ø. and Harper, D. A.: Past: paleontological statistics software package for educaton and data anlysis, Palaeontol. Electron., 4, 9 pp., 2001.
Harland, R., Asteman, I. P., and Nordberg, K.: A two-millennium dinoflagellate cyst record from Gullmar Fjord, a Swedish Skagerrak sill fjord, Palaeogeogr. Palaeoclimatol. Palaeoecol., 392, 247–260, https://doi.org/10.1016/j.palaeo.2013.09.006, 2013.
Hu, T., Zhang, J., Xu, X., Wang, X., Yang, C., Song, C., Wang, S., and Zhao, S.: Bioaccumulation and trophic transfer of antibiotics in the aquatic and terrestrial food webs of the Yellow River Delta, Chemosphere, 323, 138211, https://doi.org/10.1016/j.chemosphere.2023.138211, 2023.
Kalghatgi, S., Spina, C. S., Costello, J. C., Liesa, M., Morones-Ramirez, J. R., Slomovic, S., Molina, A., Shirihai, O. S., and Collins, J. J.: Bactericidal antibiotics induce mitochondrial dysfunction and oxidative damage in mammalian cells, Sci. Translat. Med., 5, 192ra85, https://doi.org/10.1126/scitranslmed.3006055, 2013.
Khaliq, Y. and Zhanel, G. G.: Fluoroquinolone-associated tendinopathy: a critical review of the literature, Clin. Infect. Dis., 36, 1404–1410, https://doi.org/10.1086/375078, 2003.
Lee, J. J. and Pierce, S.: Growth and physiology of Foraminifera in the laboratory; part 4 – monoxenic culture of an allogromiid with notes on its morphology, J. Protozool., 10, 404–411, https://doi.org/10.1111/j.1550-7408.1963.tb01697.x, 1963.
Lee, J. J., Sang, K., Ter Kuile, B., Strauss, E., Lee, P. J., and Faber, W. W.: Nutritional and related experiments on laboratory maintenance of three species of symbiont-bearing, large foraminifera, Mar. Biol., 109, 417–425, https://doi.org/10.1007/BF01313507, 1991.
Lienert, J., Bürki, T., and Escher, B. I.: Reducing micropollutants with source control: substance flow analysis of 212 pharmaceuticals in faeces and urine, Water Sci. Technol., 56, 87–96, https://doi.org/10.2166/wst.2007.560, 2007.
Lintner, M., Biedrawa, B., Wukovits, J., Wanek, W., and Heinz, P.: Salinity-dependent algae uptake and subsequent carbon and nitrogen metabolisms of two intertidal foraminifera (Ammonia tepida and Haynesina germanica), Biogeosciences, 17, 3723–3732, https://doi.org/10.5194/bg-17-3723-2020, 2020.
Lintner, M., Balzano, S., Keul, N., Heinz, P., Maneckie, M., Klimek, A., Wanek, W., Cyran, N., Gruber, D., Schmidt, K., Holzmann, M., Golén, J., and and Tyszka, J.: Biosorption of heavy metals by microalgae: Hazardous side effects for marine organisms, Chemosphere, 372, 144080, https://doi.org/10.1016/j.chemosphere.2025.144080, 2025.
Michael, L.: The influence of three common antibiotics on coastal benthic foraminifera: implications for culture experiments and biomonitoring, Zenodo [data set], https://doi.org/10.5281/zenodo.16925684, 2025.
Mitchell, S. M., Ullman, J. L., Teel, A. L., and Watts, R. J.: pH and temperature effects on the hydrolysis of three β-lactam antibiotics: Ampicillin, cefalotin and cefoxitin, Sci. Total Environ., 466, 547–555, https://doi.org/10.1016/j.scitotenv.2013.06.027, 2014.
Müller, W. A. and Lee, J. J.: Apparent indispensability of bacteria in foraminiferan nutrition, J. Protozool., 16, 471–478, https://doi.org/10.1111/j.1550-7408.1969.tb02303.x, 1969.
Nguyen, L. M., Nguyen, N. T. T., Nguyen, T. T. T., Nguyen, T. T., Nguyen, D. T. C., and Tran, T. V.: Occurrence, toxicity and adsorptive removal of the chloramphenicol antibiotic in water: a review, Environ. Chem. Lett., 20, 1929–1963, https://doi.org/10.1007/s10311-022-01416-x, 2022.
Nigam, R., Khare, N., and Koli, N. Y.: Growing need to study foraminifera in the laboratory culture experiments: An attempt from the central west coast of India, http://drs.nio.org/drs/handle/2264/2226 (last access: 16 February 2025), 1996.
Oluwole, A. O. and Olatunji, O. S.: Photocatalytic degradation of tetracycline in aqueous systems under visible light irridiation using needle-like SnO 2 nanoparticles anchored on exfoliated gC 3 N 4, Environ. Sci. Europe, 34, 1–14, https://doi.org/10.1186/s12302-021-00588-7, 2022.
Patel, G. and Bonomo, R. A.: Status report on carbapenemases: challenges and prospects, Exp. Rev. Anti-Infect. Ther., 9, 555–570, https://doi.org/10.1586/eri.11.28, 2011.
Pierce, S.: A comparative study of two members of the family Allogromiidae (Protozoa, Foraminiferida), New York University, https://www.proquest.com/openview/5bd6895d647975607df9e23dba9845eb/1?pq-origsite=gscholar&cbl=18750&diss=y (last access: 21 August 2025), 1965.
Polovodova Asteman, I., Filipsson, H. L., and Nordberg, K.: Tracing winter temperatures over the last two millennia using a north-east Atlantic coastal record, Clim. Past, 14, 1097–1118, https://doi.org/10.5194/cp-14-1097-2018, 2018.
Röttger, R., Irwan, A., Schmaljohann, R., and Franzisket, L.: Growth of the symbiont-bearing foraminifera Amphistegina lessonii d'Orbigny and Heterostegina depressa d'Orbigny (Protozoa). Endocytobiology, endosymbiosis and cell biology, Walter de Gruyter and Co, Berlin, 125–132, https://doi.org/10.1515/9783111385068-015, 1980.
Shaw, W. V.: [57] Chloramphenicol acetyltransferase from chloramphenicol-resistant bacteria. In Methods in enzymology, in: Vol. 43, Academic Press, 737–755, https://doi.org/10.1016/0076-6879(75)43141-x, 1975.
Tahrani, L., Van Loco, J., Ben Mansour, H., and Reyns, T.: Occurrence of antibiotics in pharmaceutical industrial wastewater, wastewater treatment plant and sea waters in Tunisia, J. Water Health, 14, 208–213, https://doi.org/10.2166/wh.2015.224, 2016.
Wang, C., Lu, Y., Sun, B., Zhang, M., Wang, C., Xiu, C., Johnson, A. C., and Wang, P.: Ecological and human health risks of antibiotics in marine species through mass transfer from sea to land in a coastal area: A case study in Qinzhou Bay, the South China sea, Environ. Pollut., 316, 120502, https://doi.org/10.1016/j.envpol.2022.120502, 2023.
Xu, D., Xiao, Y., Pan, H., and Mei, Y.: Toxic effects of tetracycline and its degradation products on freshwater green algae, Ecotoxicol. Environ. Safe., 174, 43–47, https://doi.org/10.1016/j.ecoenv.2019.02.063, 2019.
Xu, L., Zhang, H., Xiong, P., Zhu, Q., Liao, C., and Jiang, G.: Occurrence, fate, and risk assessment of typical tetracycline antibiotics in the aquatic environment: A review, Sci. Total Environ., 753, 141975, https://doi.org/10.1016/j.scitotenv.2020.141975, 2021.
Zhang, Q. Q., Ying, G. G., Pan, C. G., Liu, Y. S., and Zhao, J. L.: Comprehensive evaluation of antibiotics emission and fate in the river basins of China: source analysis, multimedia modeling, and linkage to bacterial resistance, Environ. Sci. Technol., 49, 6772–6782, https://doi.org/10.1021/acs.est.5b00729, 2015.
Zhang, Y. and Trissel, L. A.: Stability of ampicillin sodium, nafcillin sodium, and oxacillin sodium in AutoDose infusion system bags, Int. J. Pharmaceut. Compound., 6, 226–229, 2002.